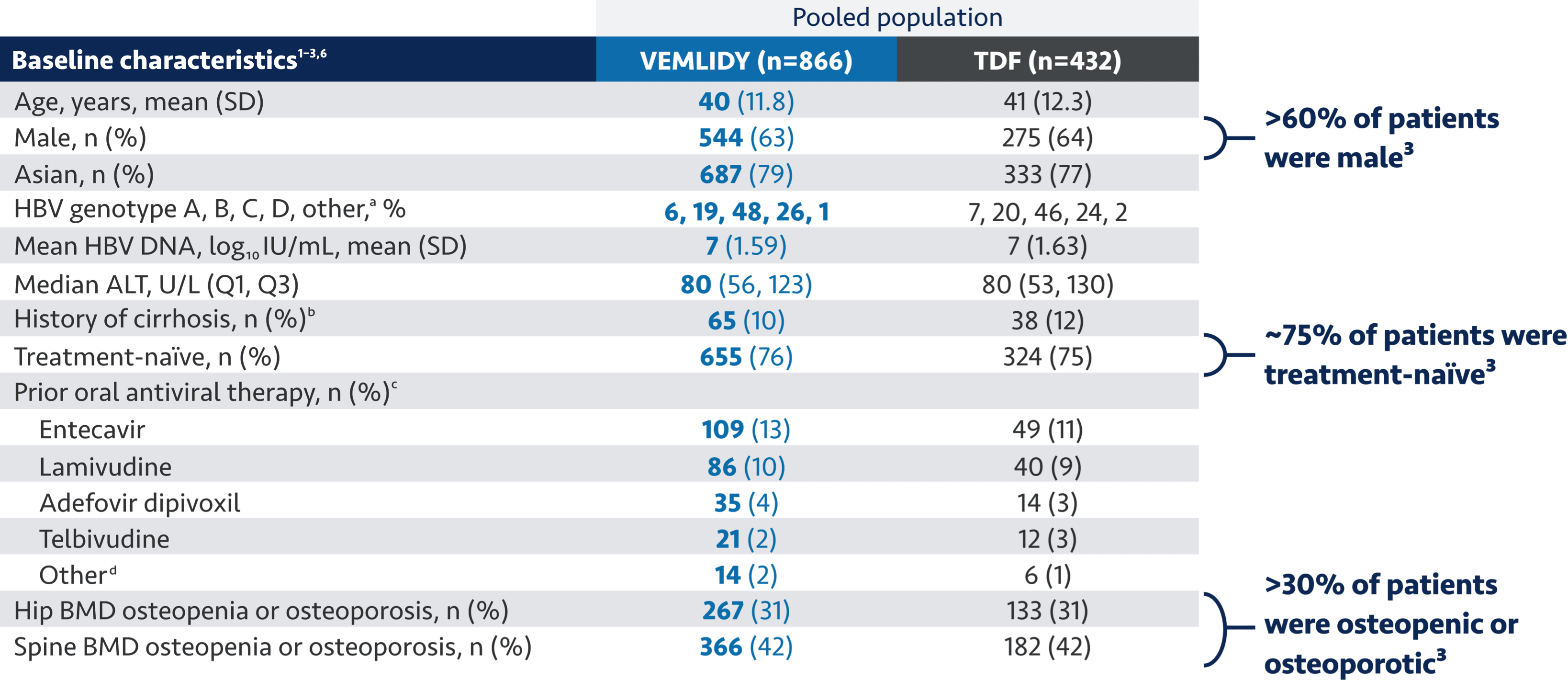
VEMLIDY is proven in robust global clinical trials1-5
*The data is not presented in the VEMLIDY label.
ALT=alanine aminotransferase.
Watch experts discuss treatment guidelines and algorithms
See how your patients could benefit from these treatment approaches
View nowHelp your patients save on prescription costs
Eligible commercially insured patients can save with the VEMLIDY Co-pay Savings Program*
*For commercially insured eligible patients only. Restrictions apply. Subject to change; click “See the details” below for full terms and conditions. This is not health insurance. Only accepted at participating pharmacies.
See the details
VEMLIDY is recommended by global treatment guidelines and algorithms as a preferred first-line therapy for the treatment of chronic hepatitis B in adults with compensated liver disease7-11,a

AASLD 2025

SABA 2022

EASL 2025

China 2022

AATA 2018
AASLD=American Association for the Study of Liver Diseases; AATA=Asian American Treatment Algorithm; EASL=European Association for the Study of the Liver; SABA=Simplified Approach Hepatitis B Algorithm.
aOther recommended first-line nucleoside/nucleotide analogs are entecavir and tenofovir disoproxil fumarate (TDF).7-11
VEMLIDY offers convenient, patient-friendly dosing with a once-daily pill1
VEMLIDY is a 25-mg pill that’s 8 mm in diameter. Pill image not to scale.
Learn moreFor your patients: Real VEMLIDY patients share their stories
Share their journeys with your patients, and download other available patient resources
See morePatient compensated by Gilead.