VEMLIDY demonstrated improvement of cirrhosis and fibrosis in chronic hepatitis B patients through 8 years1
Powerful efficacy maintained over the long term with 0% resistance1
Regression of compensated cirrhosis seen with VEMLIDY through 8 years1,2
Regression of cirrhosis in chronic hepatitis B patients with cirrhosis at baseline
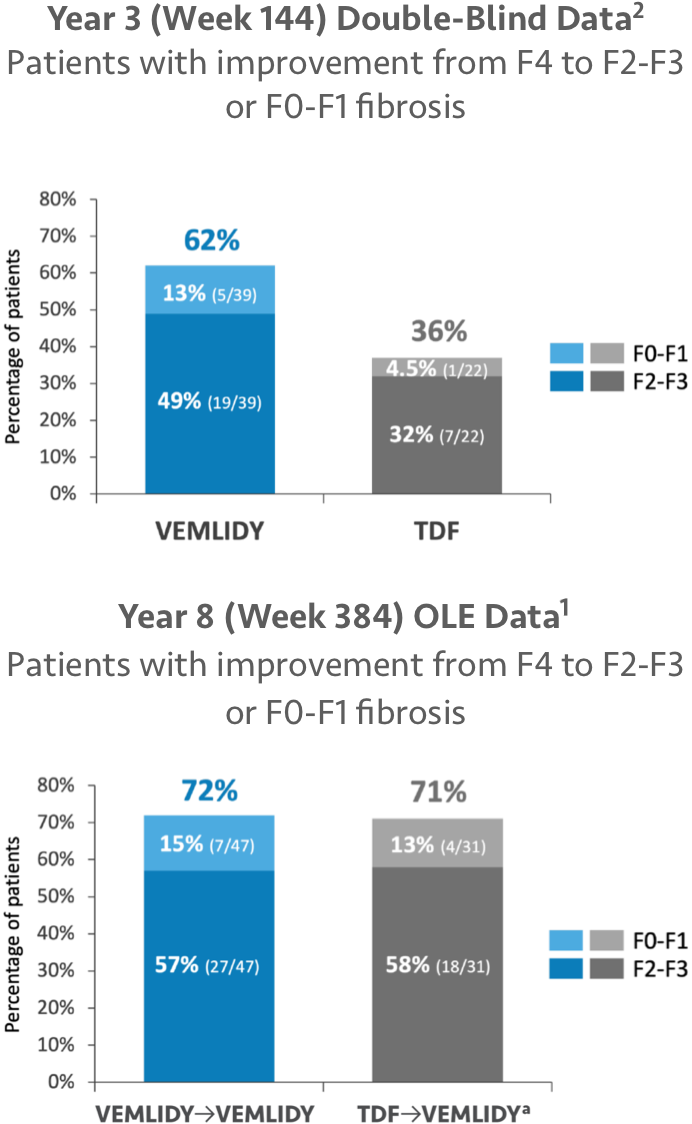
Year 3 Data: Among the 1298 randomized and treated patients, 644 remained in the double-blind phase at Week 144, and 398 patients from the VEMLIDY group and 193 patients from the TDF group had FibroTest data available for analysis at both baseline and Week 144. The graphs show the results for those patients who had F4 fibrosis (FibroTest score ≥0.75) at baseline (39 patients in the VEMLIDY group and 22 patients in the TDF group).2
Year 8 Data: Among the 1298 randomized and treated patients, 575 patients from the VEMLIDY→VEMLIDY group and 282 patients from the TDF→VEMLIDY groups had FibroTest data available for analysis at both baseline and Week 384. The graphs show the results for those patients who had F4 fibrosis (FibroTest score ≥0.75) at baseline (47 patients in the VEMLIDY→VEMLIDY group and 31 patients in the TDF→VEMLIDY groups).1
Limitations: In Trials 108 and 110 at baseline, 10% of VEMLIDY patients and 12% of TDF patients had compensated cirrhosis.1
ALT=alanine aminotransferase; OLE=open-label extension; TDF=tenofovir disoproxil fumarate.
aIncluded data from 11 patients who switched from TDF to VEMLIDY at Week 96 and 20 patients who switched from TDF to VEMLIDY at Week 144.2
Additional context regarding the data presented above
- Change from baseline in fibrosis assessed by FibroTest score (missing=excluded analysis) for VEMLIDY vs TDF was a secondary endpoint in Trials 108 and 110. Liver biopsies and FibroScan® tests were not conducted as part of Trials 108 and 1103,4
- FibroTest is a noninvasive measure of liver fibrosis and combines 5 standard biomarkers: gamma-glutamyl transpeptidase, total bilirubin, alpha-2-macroglobulin, apolipoprotein A1, and haptoglobin. Note that FibroTest does not include ALT. FibroTest has been validated for assessing fibrosis in patients with chronic hep B5
- The clinical relevance of these changes in FibroTest scores is not known
- This analysis is not presented in the VEMLIDY full Prescribing Information
- Cirrhosis analysis is not powered for statistical significance, and data should be considered descriptive only
