More of your patients with chronic HBV infection may be eligible for treatment1
Patients who are HBsAg positive and have detectable HBV DNA levels should not be overlooked, regardless of ALT levels.
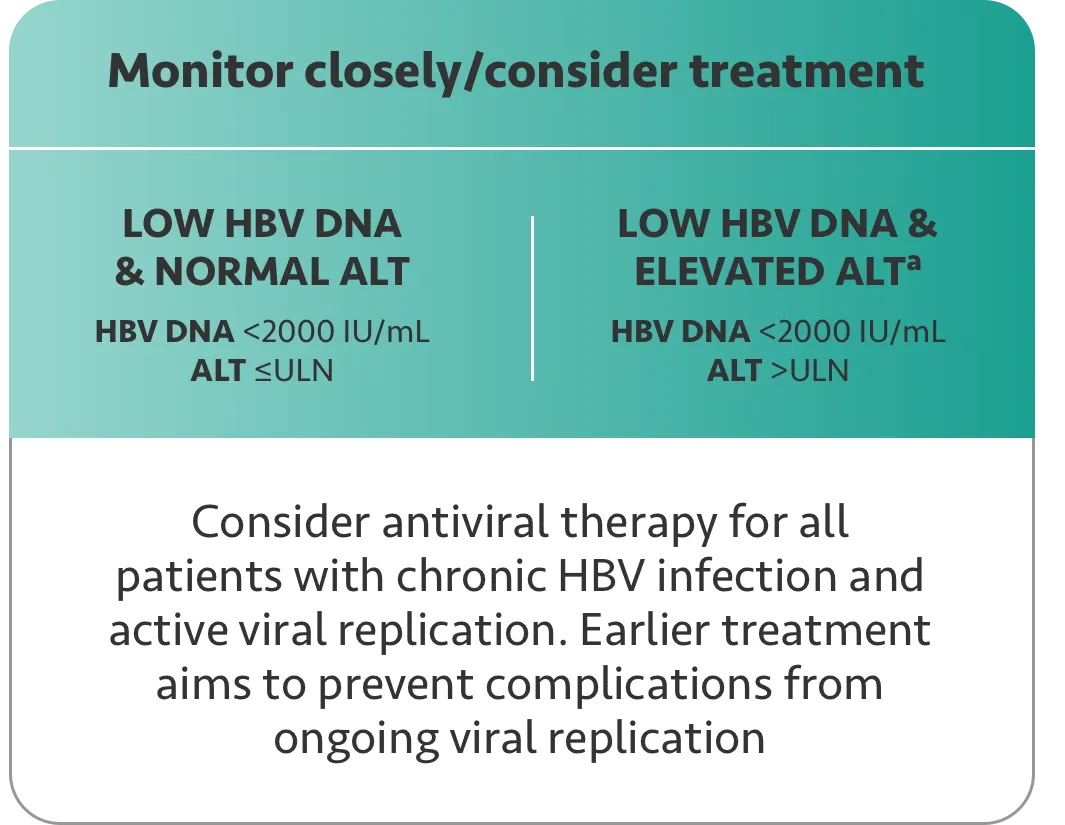
Expanded treatment criteria include patients with elevated HBV DNA and normal ALT or low HBV DNA and elevated ALT.
Tap each section to see the recommendations.
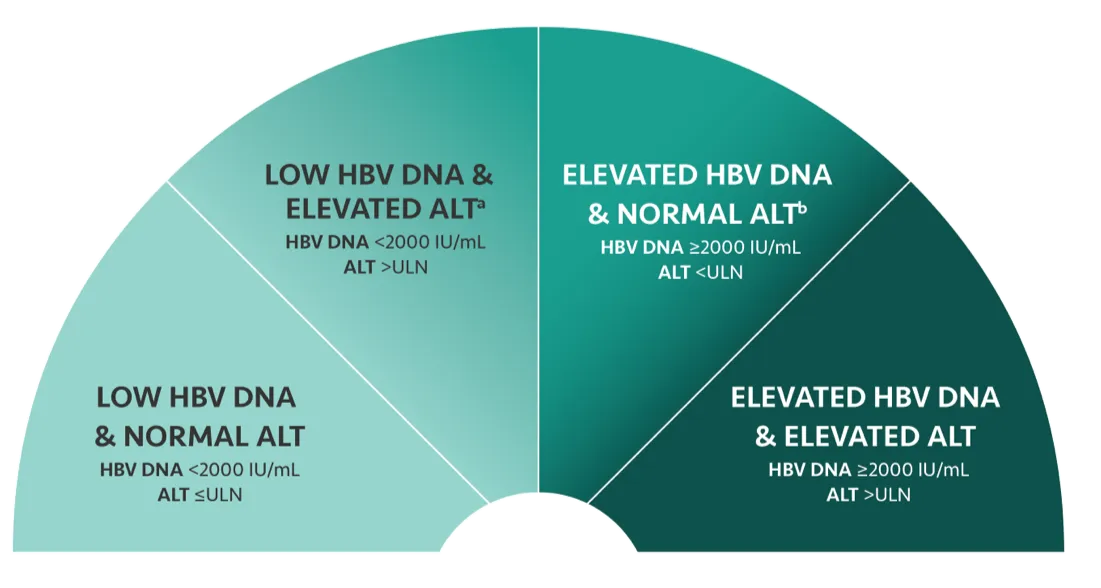

Prioritization of treatment depends on a patient’s risk of disease progression and HCC, as indicated by virological and host factors.
a Patients with persistently low HBV DNA (<2000 IU/mL) and persistently elevated ALT (>ULN) can be treated. However, consider other liver diseases that could also be implicated.
b Treatment is recommended if HBV DNA is ≥2000 IU/mL and if patient has any of the following: ALT >ULN, fibrosis, cirrhosis, risk factors for HCC, extrahepatic manifestations, immunosuppression, or risk for HBV transmission.
A 2024 overview of clinical evidence conducted by experts in the US and Taiwan showed that patients with low HBV DNA/elevated ALT or elevated HBV DNA/normal ALT remain at risk for disease progression, including the development of HCC.2
(Mak LY, et al, 2024)
ALT=alanine aminotransferase; HBsAg=hepatitis B surface antigen; HBV=hepatitis B virus; HCC=hepatocellular carcinoma; ULN=upper limit of normal.

38% of patients were classified into an indeterminate/gray zone where treatment intervention is not clearly recommended3
The 2015 study by Di Bisceglie et al included 2290 adult participants from a Hepatitis B Research Network Cohort or Treatment study. A total of 1571 participants met inclusion criteria and 1390 participants had sufficient data available to allow a baseline phenotype to be determined. This cohort was enrolled in 19 different medical sites in the US and 1 site in Canada. Within this cohort, the largest single phenotype (38%; 524/1390) was classified as "indeterminate" when using a fixed ALT ULN of 30 U/L for men and 20 U/L for women.3
