VEMLIDY: A demonstrated renal safety profile through 8 years1,2
Across multiple trials, VEMLIDY demonstrated a reduced effect on renal safety parameters and improved renal function when compared to baseline1,2
VEMLIDY showed reduced impact on renal safety parameters at Week 144
~75% of patients enrolled in pivotal trials 108/110 were treatment-naïve.2
Renal effects of VEMLIDY and TDF were compared in a pooled analysis of Trials 108 and 1101
Median baseline eGFRCG was 106 mL/min and 105 mL/min for VEMLIDY and TDF, respectively.1
Change in eGFRCG from baseline at Week 144 (pooled)3
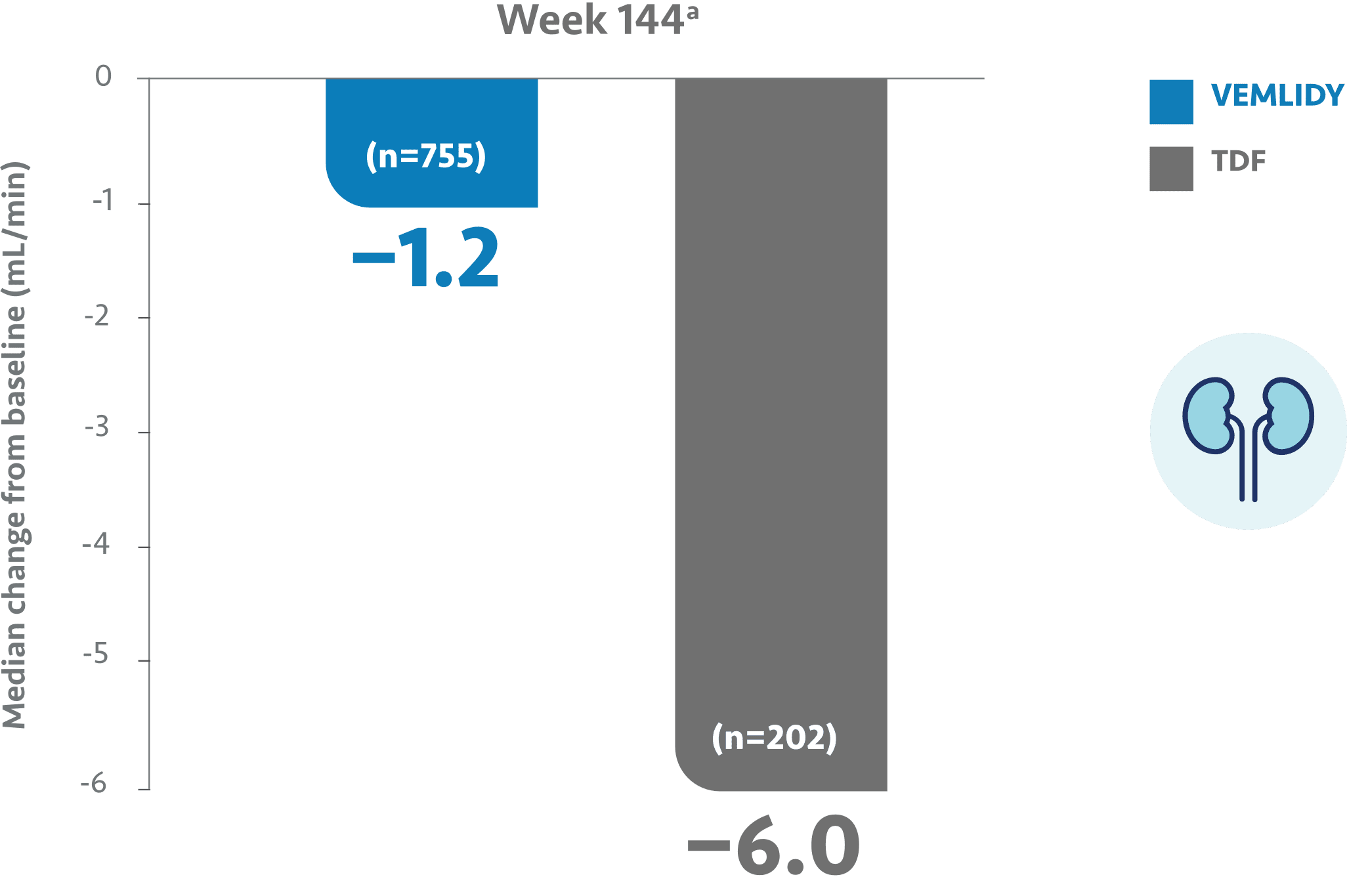
- In adult patients with chronic hepatitis B, the mean increase in serum creatinine was <0.1 mg/dL in both treatment groups at both Week 96 and Week 1441,3
- The median change in eGFRCG from baseline was smaller for VEMLIDY vs TDF3
- Median change from baseline to Week 96 in eGFRCG was -1.2 mL/min in the VEMLIDY group (n=790) and -4.8 mL/min in those receiving TDF (n=390)1-3
a The Week 144 analysis did not include the 180 patients (HBeAg⎻: 66 patients; HBeAg+: 114 patients) who had rolled over from double-blind TDF to open-label VEMLIDY at Week 96 prior to the trial amendment.3
The long-term clinical significance of these renal laboratory changes on adverse reaction frequencies between VEMLIDY and TDF is not known.1
- For patients with mild, moderate, or severe renal impairmentb
- For patients with ESRDc who are receiving chronic hemodialysis
- On days of hemodialysis, administer VEMLIDY after completion of hemodialysis treatment
- VEMLIDY is not recommended in patients with ESRD who are not receiving chronic hemodialysis
eGFRCG=estimated glomerular filtration rate by Cockcroft-Gault method, also referred to as eCrCl (estimated creatinine clearance); TDF=tenofovir disoproxil fumarate.
beCrCl ≥15 mL/min.2
ceCrCl <15 mL/min.2
Long-term renal safety parameters remained stable through 8 years in patients taking VEMLIDY
Pooled safety analysis (Week 384): Pooled safety analysis (observed data) from Trials 108 and 110 was assessed at Week 384. This analysis included 866 patients who initiated VEMLIDY at baseline, 207 patients who switched from TDF to VEMLIDY at Week 96, and 225 patients who switched from TDF to VEMLIDY at Week 144.2,a
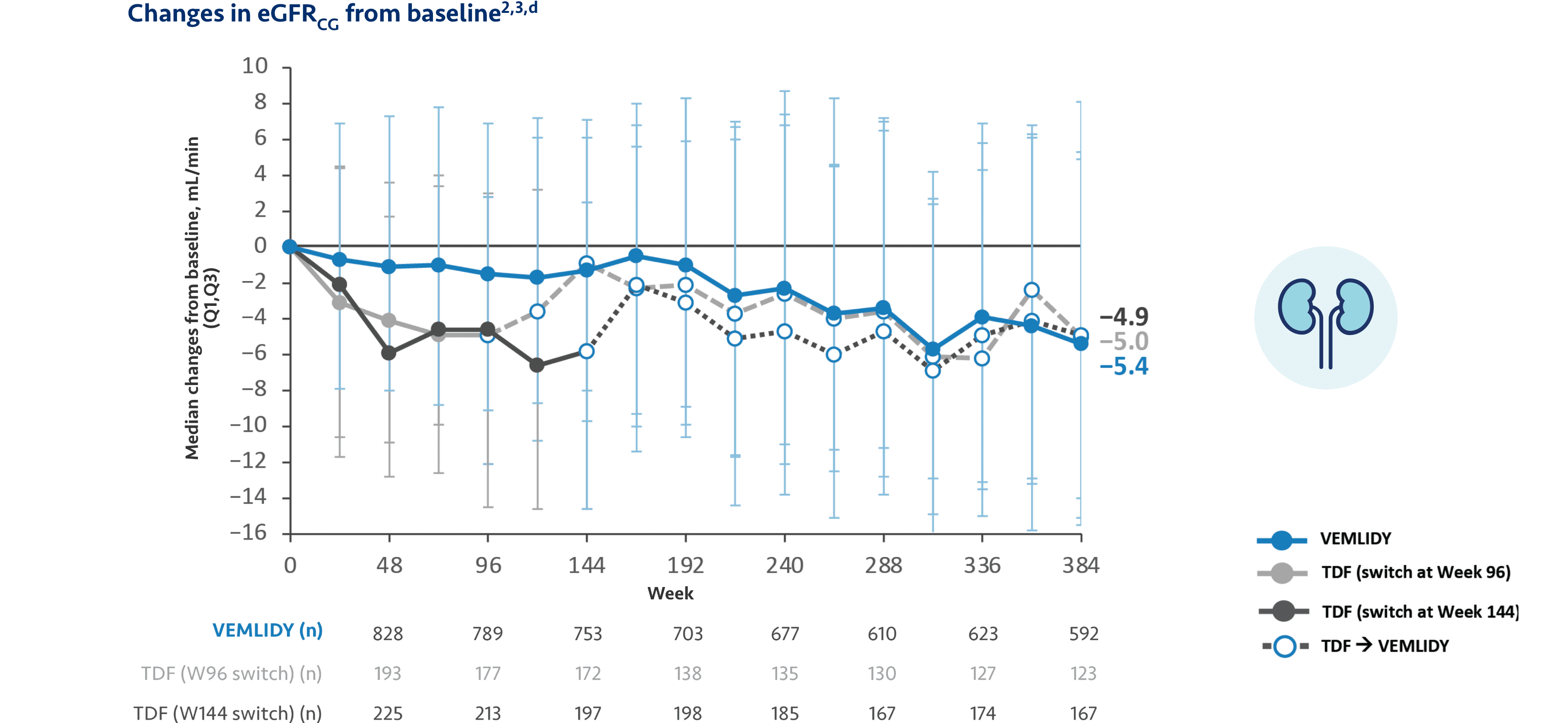
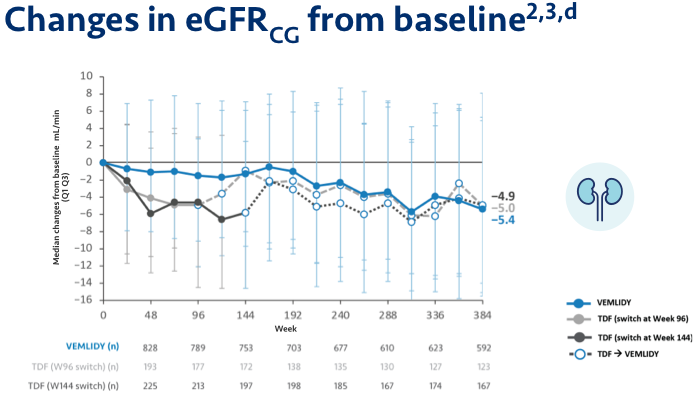
Median change in eGFRCG from Week 96 to 120: -0.6 mL/min in patients who remained on VEMLIDY and +1.8 mL/min in patients who switched from TDF to VEMLIDY.1
The long-term clinical significance of these renal laboratory changes on adverse reaction frequencies between VEMLIDY and TDF is not known.1
Most common adverse reactions (incidence ≥5%; all grades) at Week 384 open-label extension were headache, upper respiratory tract infection, nasopharyngitis, arthralgia, hypertension, cough, and back pain.2
The 8-year analysis is not presented in the VEMLIDY full Prescribing Information.
d VEMLIDY group includes VEMLIDY patients who rolled over to open-label VEMLIDY at Week 96 or Week 144.
Warnings and Precautions
- New Onset or Worsening Renal Impairment: Postmarketing cases of renal impairment, including acute renal failure, proximal renal tubulopathy (PRT), and Fanconi syndrome have been reported with TAF-containing products. Patients with impaired renal function and/or taking nephrotoxic agents (including NSAIDs) are at increased risk of renal-related adverse reactions. Discontinue VEMLIDY in patients who develop clinically significant decreases in renal function or evidence of Fanconi syndrome. Monitor renal function in all patients – See Dosage and Administration.
