VEMLIDY—proven long-term viral suppression in chronic hepatitis B patients with compensated liver disease1,2
Powerful efficacy maintained over the long term with 0% resistance2
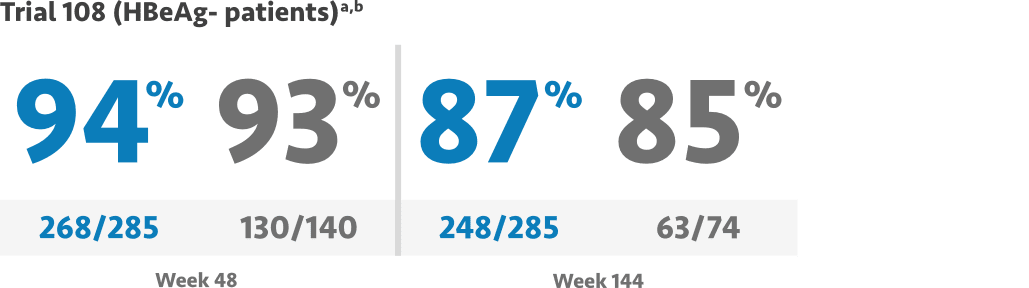
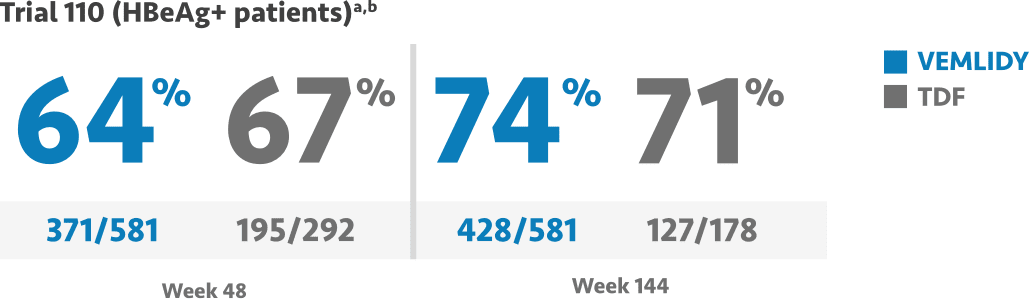
VEMLIDY demonstrated powerful antiviral efficacy with viral suppression at Weeks 48, 96, and 144 (HBV DNA <29 IU/mL)1-6
~75% of patients enrolled in pivotal Trials 108/110 were treatment-naïve.2

Mean baseline plasma HBV DNA was 5.8 log10 IU/mL in Trial 108 and 7.6 log10 IU/mL in Trial 110.1
- Trial 108 viral suppression at Week 96: VEMLIDY 90% (257/285), TDF 91% (127/140)3
- Trial 110 viral suppression at Week 96: VEMLIDY 73% (423/581), TDF 75% (218/292)4
Primary efficacy endpoint: The proportion of patients with HBV DNA <29 IU/mL and noninferiority to TDF (10% margin; 95% CI approach) at Week 48 for both trials.1,4,6
Most common adverse reactions (incidence ≥5%; all grades) in clinical studies through Week 144 were headache, upper respiratory tract infection, abdominal pain, cough, back pain, arthralgia, fatigue, nausea, diarrhea, dyspepsia, and pyrexia.1,4,6,7
CI=confidence interval; HBeAg=hepatitis B envelope antigen; TDF=tenofovir disoproxil fumarate.
aPatient populations analyzed included all treatment-naïve and treatment-experienced patients who were randomized into the trial and received at least 1 dose of study drug; a missing=failure approach was used.1
bThe Week 144 analysis did not include the 66 patients from the TDF group in Trial 108 and the 114 patients from the TDF group in Trial 110 who had rolled over from double-blind TDF to open-label VEMLIDY at Week 96 prior to the study amendment.7
Long-term viral suppression with VEMLIDY through 8 years
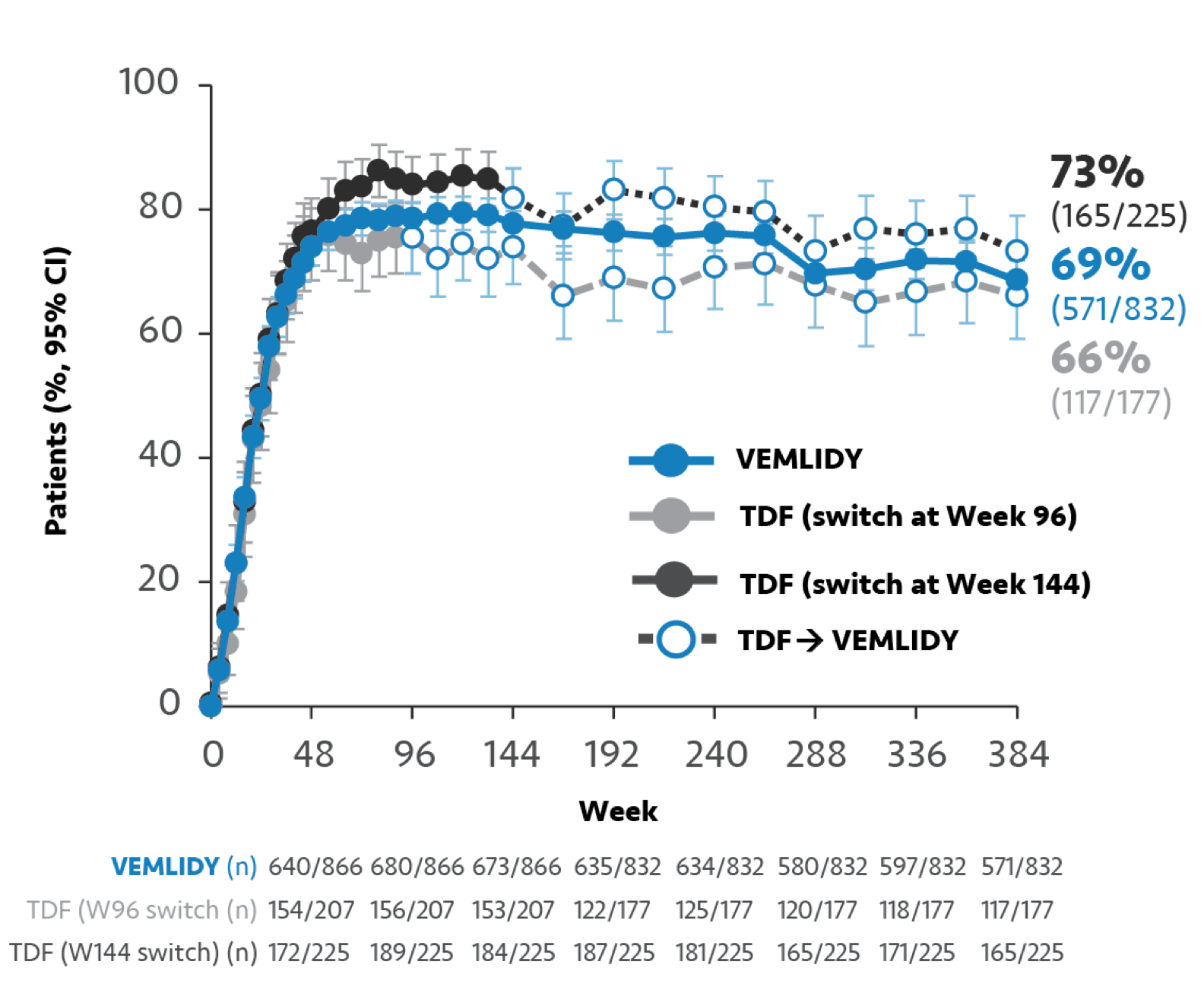
Pooled Week 384 analysis: Pooled efficacy analysis from Trials 108 and 110 was assessed at Week 384 analysis for patients in the full analysis set. This analysis included 866 patients who continued on VEMLIDY (pooled), 207 patients who switched from TDF to VEMLIDY at Week 96, and 225 patients who switched from TDF to VEMLIDY at Week 144.2,c,d
Missing=Failure (M=F) analysis from Trials 108/110:
HBV DNA <29 IU/mL at Year 82,7
Missing=Excluded (M=E) analysis from Trials 108/110:
HBV DNA <29 IU/mL at Year 82,7
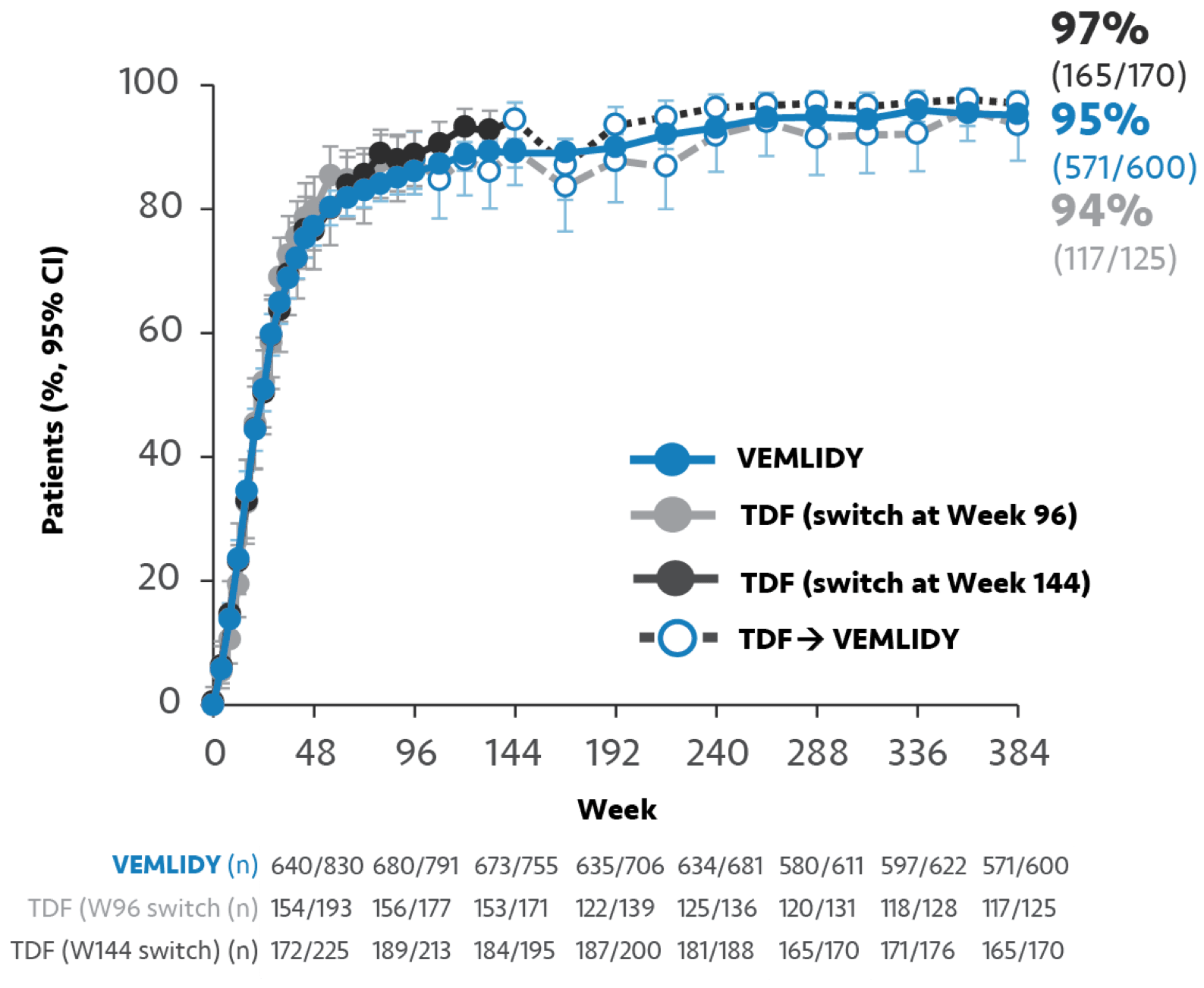
Due to early study discontinuations, which were mainly not attributable to lack of efficacy or AEs, an M=E approach was performed. In the M=E approach, all missing data were excluded in the computations from this analysis.
Limitations: The M=E data are not powered to show statistical significance and should be considered as descriptive only. Neither the M=E data nor the 8-year data are presented in the VEMLIDY full Prescribing Information.
In an M=E analysis, any patients with missing data are excluded from the final analysis. This approach assumes that the missing data are random and not related to treatment outcomes.
In an M=F analysis, missing values are included. This approach accounts for missing data points as a “failure” in the final analysis.
cMean baseline plasma HBV DNA: 5.8 log10 IU/mL in Trial 108 and 7.6 log10 IU/mL in Trial 110.1
dOne site did not participate in Protocol amendment 3, and all patients from this site (n=64) discontinued the study on or before Year 3 (Week 144). Therefore, those patients who completed the planned study treatments were excluded from the M=F analysis for all visits after Year 3.2
In Trials 108/110, genotypic resistance analysis was performed on patients experiencing either1:
- Virologic breakthrough (2 consecutive visits with HBV DNA ≥69 IU/mL [400 copies/mL] after having been <69 IU/mL, or ≥1.0-log10 increase in HBV DNA from nadir)
- Early discontinuation at or after Week 24 with HBV DNA ≥69 IU/mL
Warnings and Precautions
- Risk of Development of HIV-1 Resistance in HBV/HIV-1 Coinfected Patients: Due to this risk, VEMLIDY alone should not be used for the treatment of HIV-1 infection. Safety and efficacy of VEMLIDY have not been established in HBV/HIV-1 coinfected patients. HIV antibody testing should be offered to all HBV-infected patients before initiating therapy with VEMLIDY, and, if positive, an appropriate antiretroviral combination regimen that is recommended for HBV/HIV-1 coinfected patients should be used.
