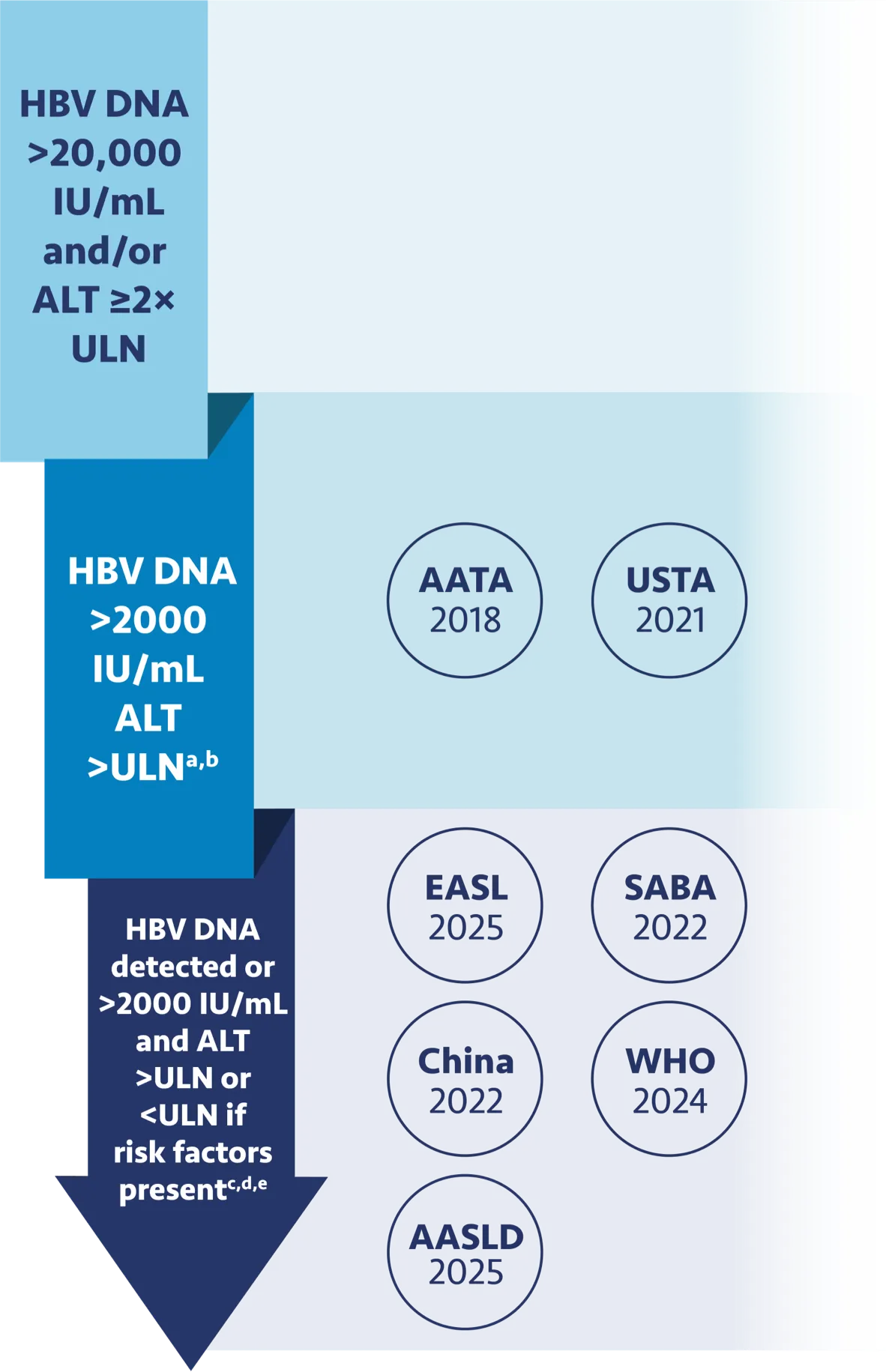
There’s clear momentum across the globe to treat chronic hepatitis B earlier1
Over time, expert groups have expanded treatment criteria, lowering thresholds for HBV DNA and ALT1-7
In a Korean HBV population modeling study, the estimated treatment-eligible patient population increased from
25%
54%8,f
Based on modeling estimates of 262,000/1,210,000 patients (UI: 994,000-1,206,000) for current treatment criteria and 676,000/1,210,000 patients (UI: 994,000-1,206,000) for expanded treatment criteria.
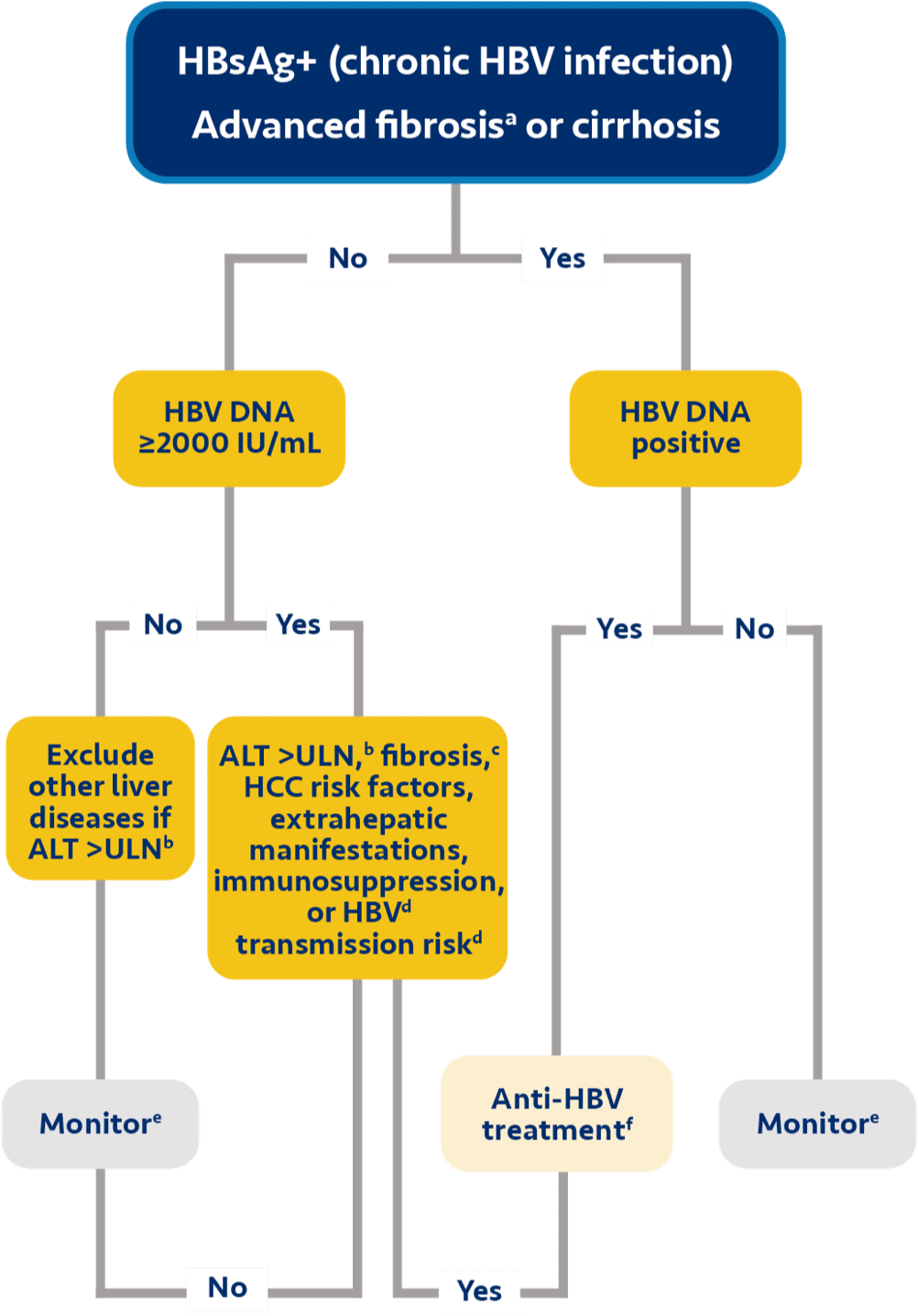
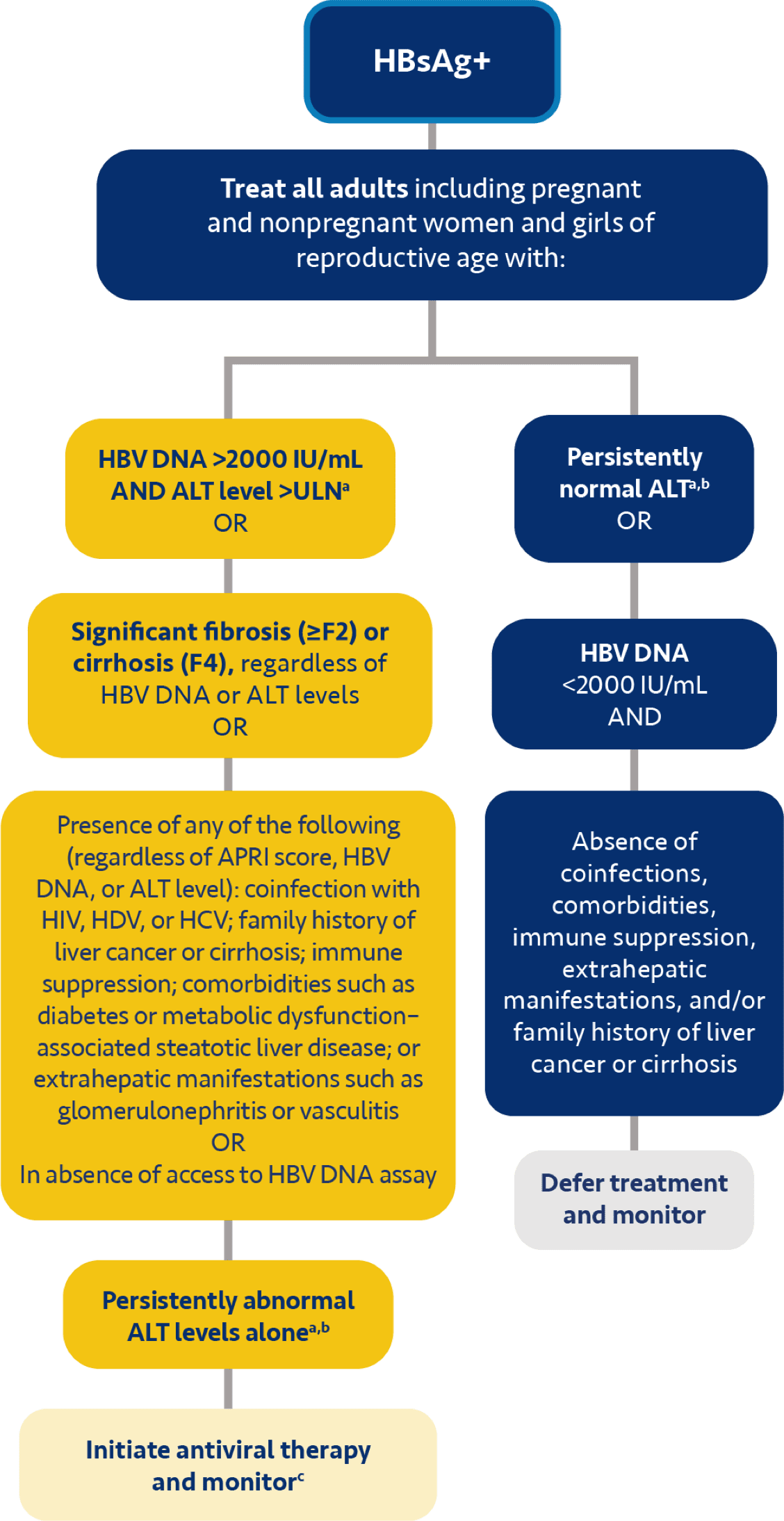
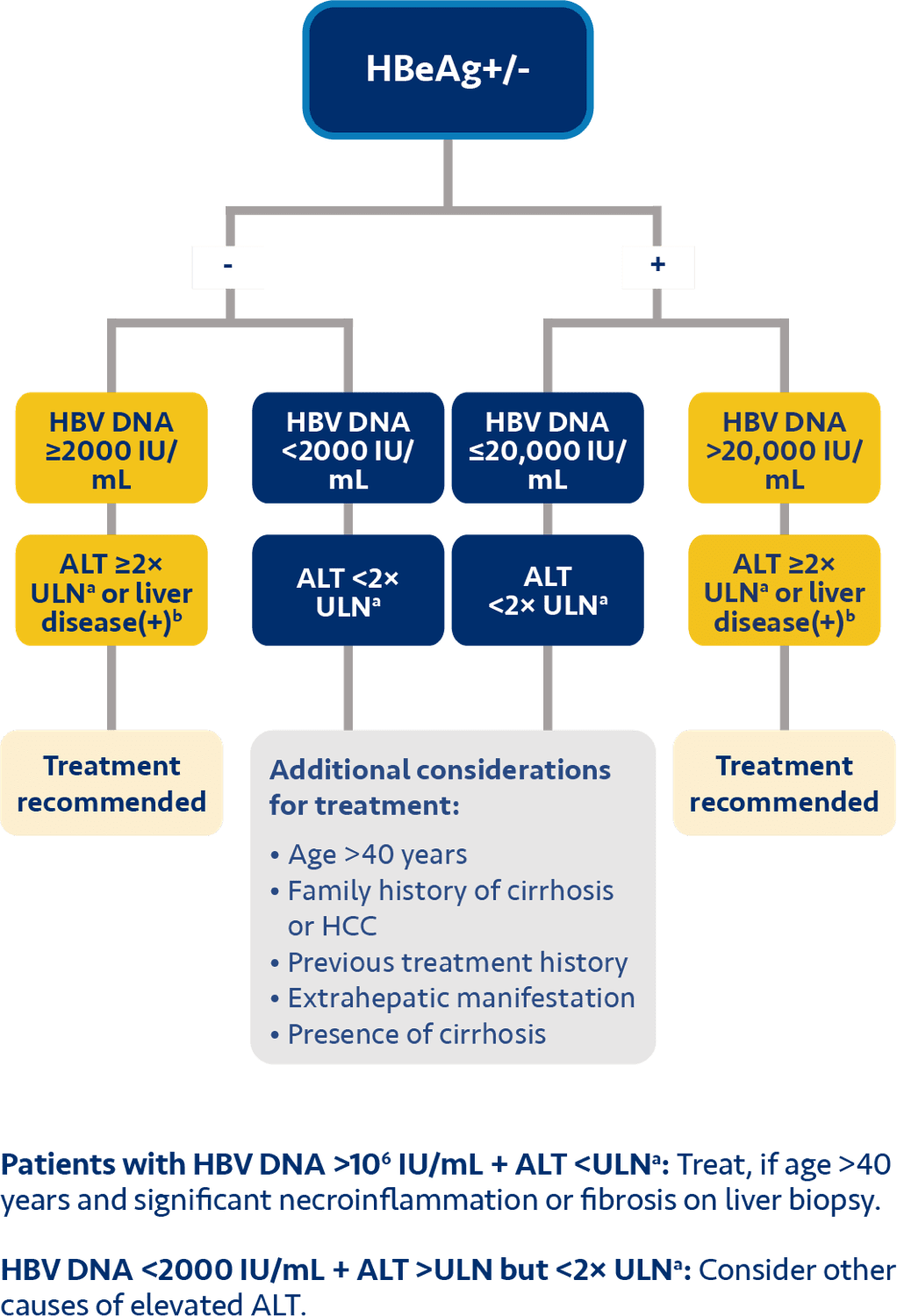
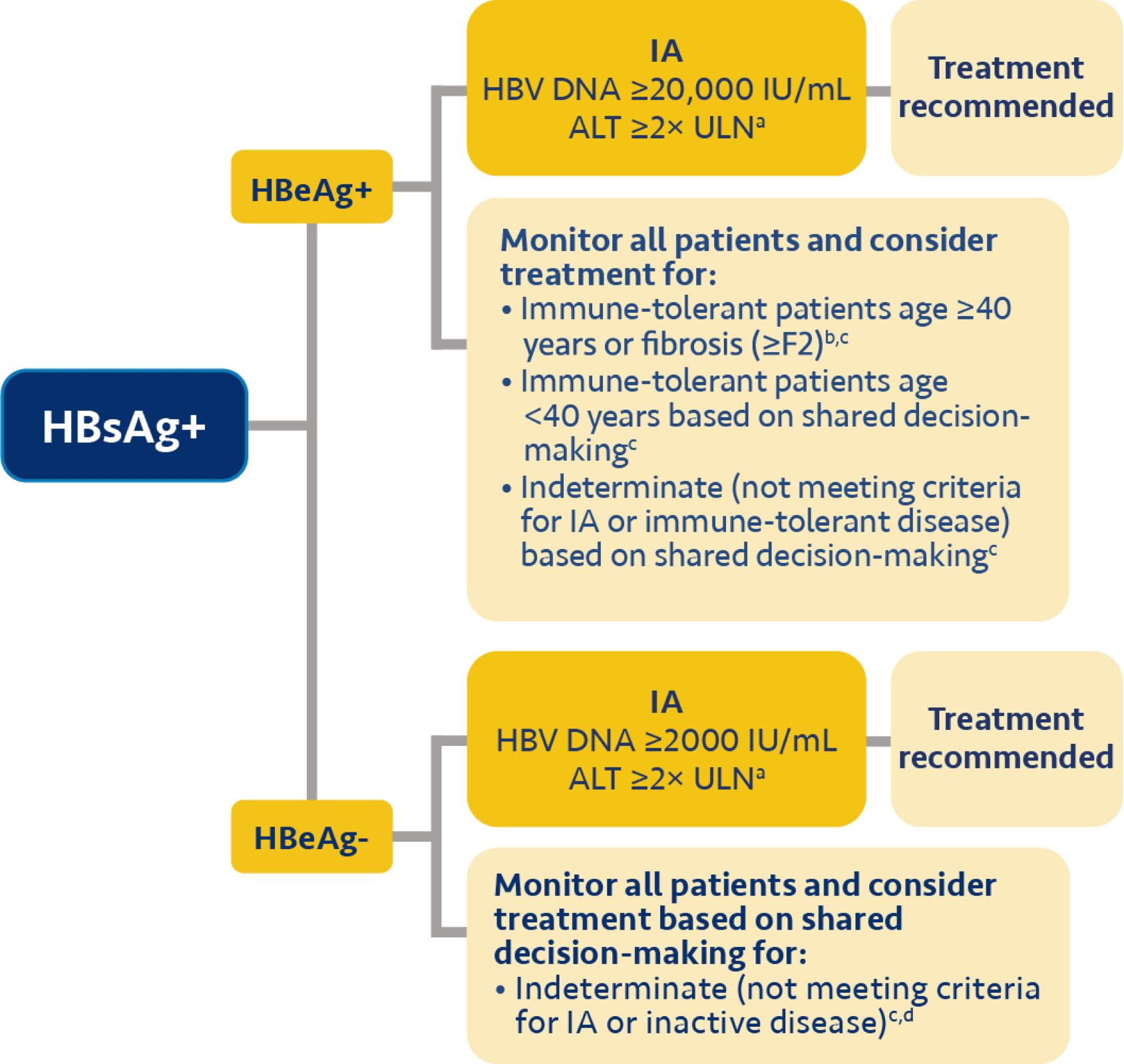
a AATA recommends treatment for HBeAg+/- if ALT >ULN or if fibrosis (≥F2)/other risk factors are present.2
b USTA recommends considering treatment based on risk factors for developing HCC, as well as patient's age, lifestyle, and desire to undergo treatment.3
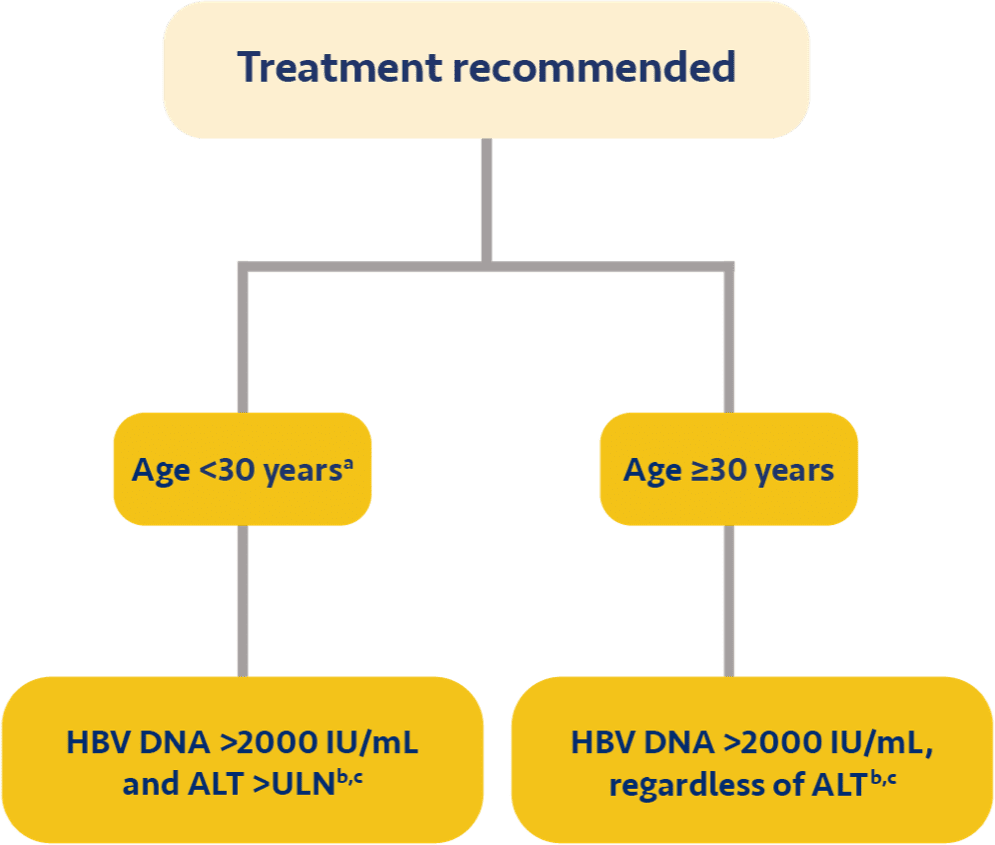
c SABA recommends treatment for patients <30 years old if they have HBV DNA >2000 IU/mL and ALT >ULN.4
d The China guidelines recommend considering treatment for people with a family history of HBV-related cirrhosis or HCC; age >30 years; or HBV-related extrahepatic manifestations.6
e For HBeAg+, AASLD recommends considering treatment for patients with HBV DNA >10 million IU/mL, ALT ≤ULN, and age ≥40 years or fibrosis (≥F2), or age <40 years based on shared decision-making; or for patients with HBV DNA ≥20,000 to ≤10 million IU/mL and ALT <2× ULN, or HBV DNA >10 million IU/mL and ALT 1-<2× ULN, based on shared decision-making. For HBeAg-, AASLD recommends considering treatment based on shared decision-making for patients with HBV DNA ≥2000 IU/mL and ALT <2× ULN or patients with HBV DNA <2000 IU/mL and ALT >ULN.7
f An estimate of untreated patients with chronic hep B who would be eligible for treatment per outlined criteria. Lim et al populated a fully dynamic transmission and Markov disease burden model with historical Korean-specific background population, mortality, and epidemiologic HBV data to track the distribution of HBsAg across sex, age, year, disease stage, and viral load. Using the PRoGReSs model, 4 scenarios were examined to assess the future impact of different treatment rates and eligibility requirements compared with the current treatment paradigm. The study was funded in part by Gilead Sciences, Inc.8

VEMLIDY is a preferred first-line therapy for the treatment of chronic hepatitis B in adults with compensated liver disease, as recommended by multiple treatment guidelines/algorithms1-7,g
Select one of the treatment guidelines/algorithms to see additional details.
The 2022 SABA guideline was funded by Gilead Sciences, Inc., and developed independently by the SABA panel.
The development of the 2018 AATA was supported, in part, by an independent grant from Gilead Sciences, Inc.

AASLD=American Association for the Study of Liver Diseases; AATA=Asian American Treatment Algorithm; AFP=alpha-fetoprotein; ALT=alanine aminotransferase; EASL=European Association for the Study of the Liver; HBeAg=hepatitis B envelope antigen; HBsAg=hepatitis B surface antigen; HBV=hepatitis B virus; HCC=hepatocellular carcinoma; SABA=Simplified Approach Hepatitis B Algorithm; TDF=tenofovir disoproxil fumarate; UI=uncertainty interval; ULN=upper limit of normal; USTA=United States Treatment Algorithm; WHO=World Health Organization.
g Other recommended first-line nucleoside/nucleotide analogs are entecavir and TDF.1-7
h ULN criteria for men and women, respectively: AASLD 2025: 35 U/L and 25 U/L; AATA 2018: local laboratory range; China 2022: unspecified; EASL 2025: 40 U/L and 40 U/L; SABA 2022: 30 U/L and 19 U/L; USTA 2021: 30 U/L and 19 U/L; WHO 2024: 30 U/L and 19 U/L.1-7
i For HBeAg+, AASLD recommends considering treatment for patients with HBV DNA >10 million IU/mL, ALT ≤ULN, and age ≥40 years or fibrosis (≥F2), or age <40 years based on shared decision-making; or for patients with HBV DNA ≥20,000 to ≤10 million IU/mL and ALT <2× ULN, or HBV DNA >10 million IU/mL and ALT 1-<2× ULN, based on shared decision-making. For HBeAg-, AASLD recommends considering treatment based on shared decision-making for patients with HBV DNA ≥2000 IU/mL and ALT <2× ULN or patients with HBV DNA <2000 IU/mL and ALT >ULN.7
j Liver disease assessed by noninvasive testing or liver biopsy showing significant liver inflammation (≥A2) or fibrosis (≥F2).2,7
k SABA recommends treatment for patients <30 years old if they have HBV DNA >2000 IU/mL and ALT >ULN.4
l The China guidelines recommend considering treatment for people with a family history of HBV-related cirrhosis or HCC; age >30 years; or HBV-related extrahepatic manifestations.6
m USTA recommends considering treatment based on risk factors for developing HCC, as well as patient’s age, lifestyle, and desire to undergo treatment.3
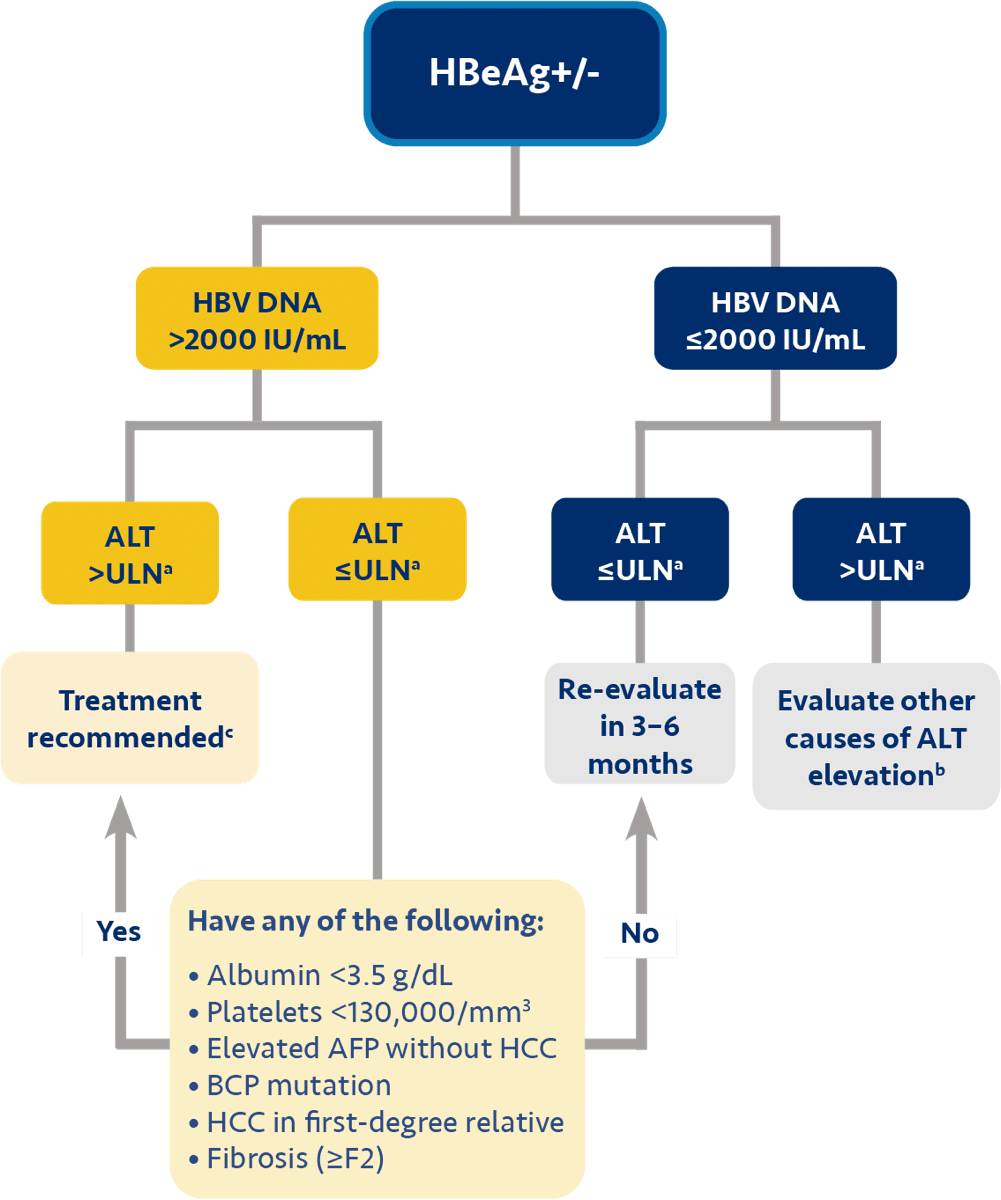
n Albumin <3.5 g/dL, platelet count <130,000/mm3, presence of basal core promoter mutation, HCC in first-degree relative, or elevated AFP in the absence of HCC.2
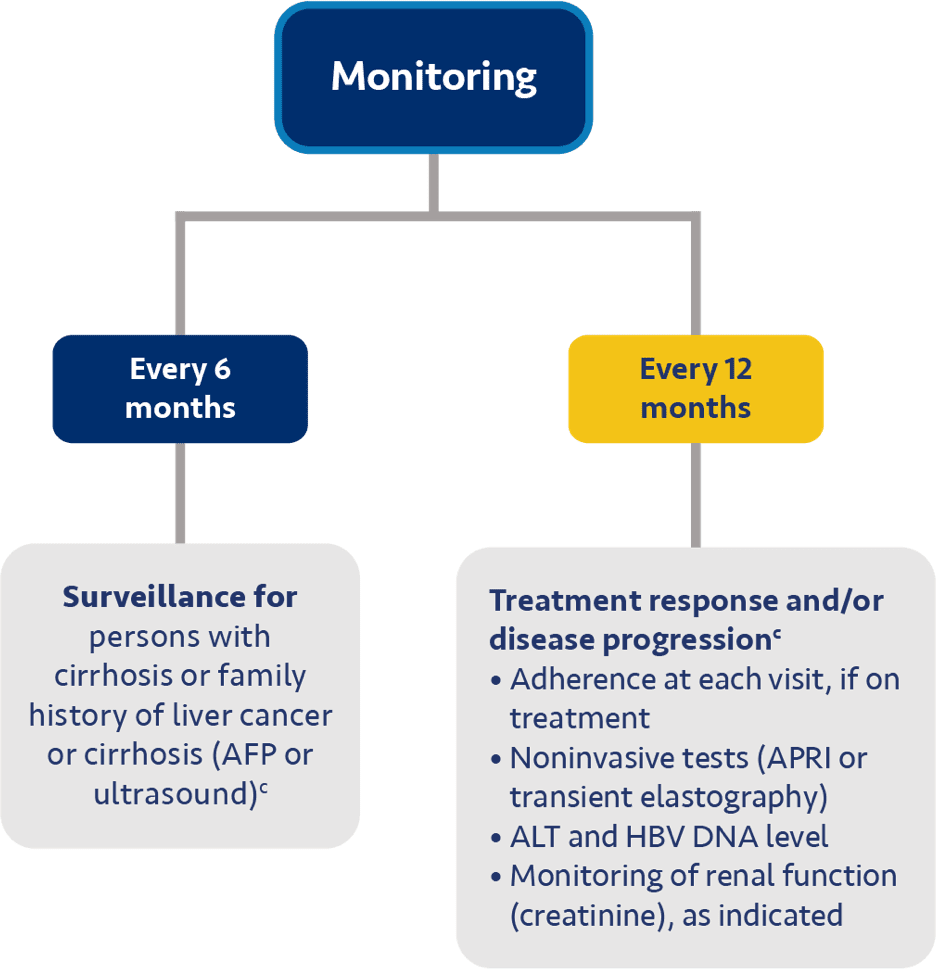
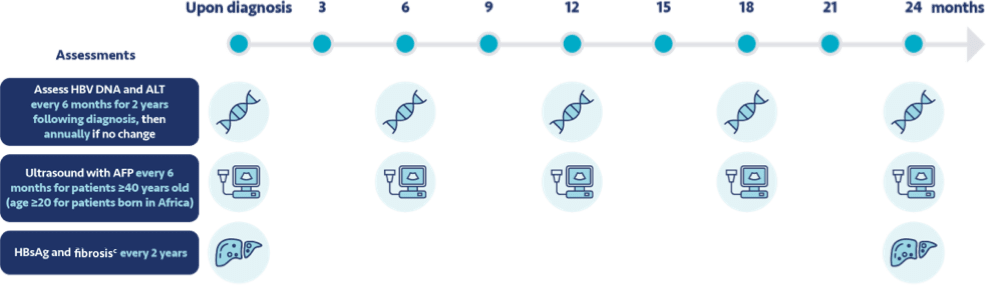
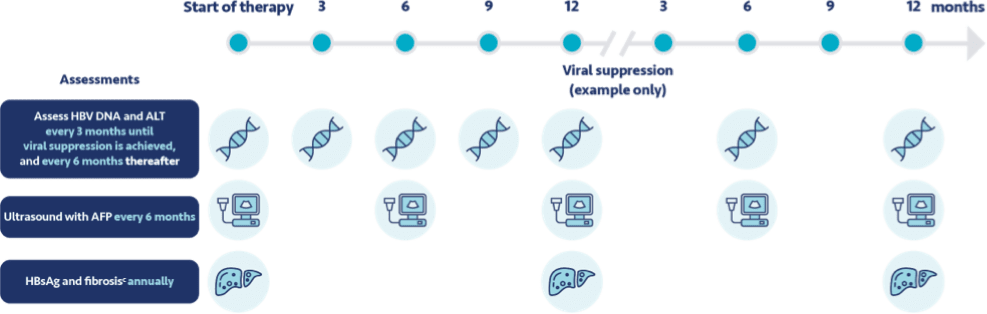
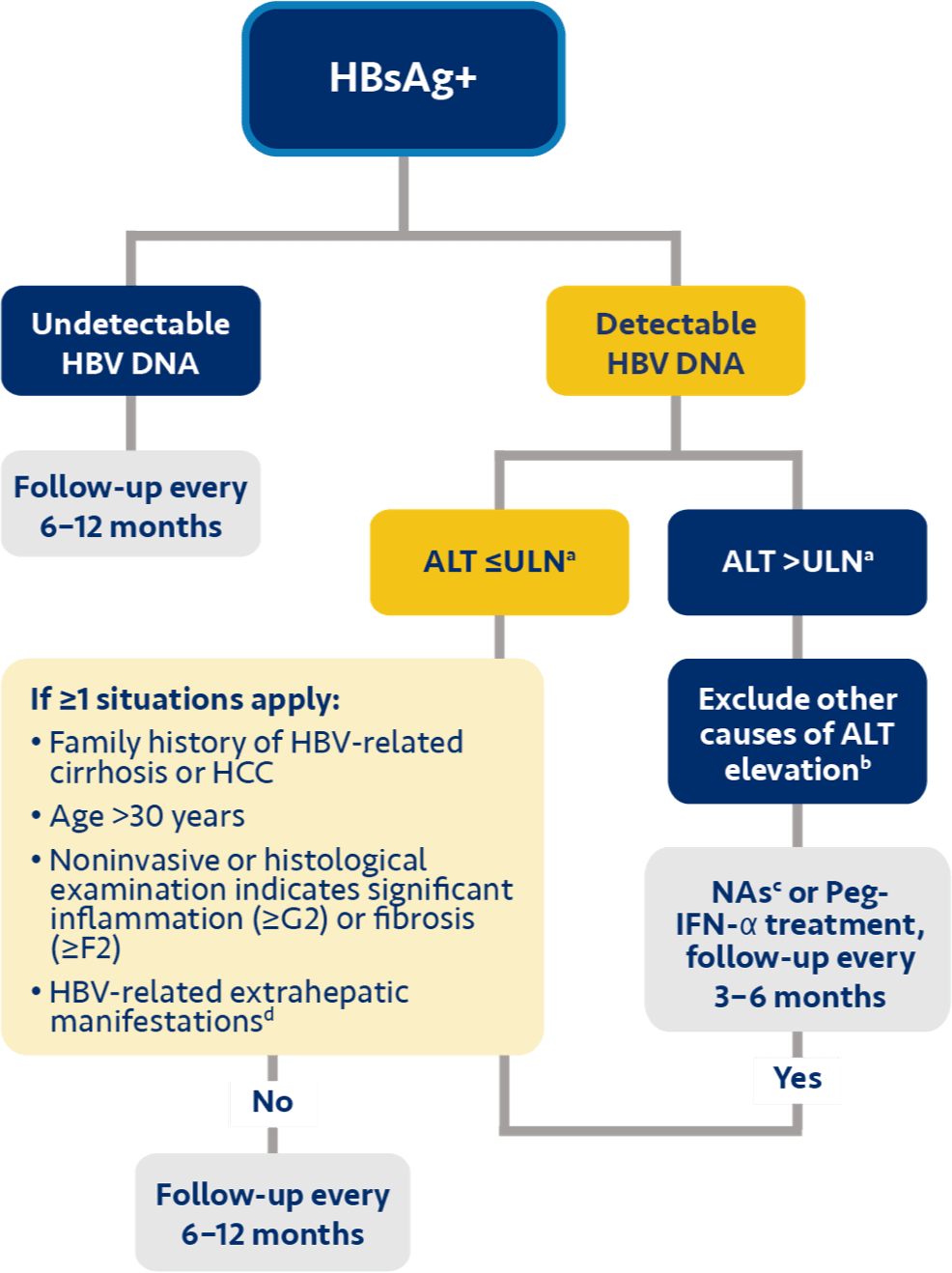
Guideline-based recommendations for chronic hepatitis B management in specific patient populations

This chart does not include the complete prescribing and dosing considerations for using these medications. Please refer to the full Prescribing Information for each medication. Comparison of drugs does not imply comparable clinical effectiveness, safety, or tolerability. Individual prescribing decisions should be made at the discretion of the provider.
eGFR=estimated glomerular filtration rate; NSAID=nonsteroidal anti-inflammatory drug; TAF=tenofovir alafenamide.
o VEMLIDY is not recommended in patients with end stage renal disease (ESRD; estimated creatinine clearance [eCrCl] <15 mL/min) who are not receiving chronic hemodialysis; in patients on chronic hemodialysis, on hemodialysis days, administer VEMLIDY after completion of hemodialysis treatment.9
p Postmarketing cases of renal impairment, including acute renal failure, proximal renal tubulopathy (PRT), and Fanconi syndrome have been reported with TAF-containing products. Patients with impaired renal function and/or taking nephrotoxic agents (including NSAIDs) are at increased risk of renal-related adverse reactions. Discontinue VEMLIDY in patients who develop clinically significant decreases in renal function or evidence of Fanconi syndrome. Monitor renal function in all patients. See Dosage and Administration.9
q VEMLIDY and entecavir are preferred in chronic hepatitis B patients with deteriorating renal function or low eGFR and/or osteopenia/osteoporosis, particularly in older age. Per EASL, VEMLIDY is preferred to entecavir in patients with previous exposure to lamivudine.5,7
r Generic TDF and generic entecavir are therapeutically equivalent to the respective brand name drug. These generic products are designated Therapeutic Equivalence Code AB, which the FDA considers therapeutically equivalent to other pharmaceutically equivalent products. Please refer to the appropriate manufacturers of generic TDF and generic entecavir for the full Prescribing Information.
s TDF 300 mg does not require dosing adjustment in patients with eCrCl ≥50 mL/min. For patients with eCrCl 30-49 mL/min, 10-29 mL/min, and ESRD on chronic hemodialysis, the dosing is every 48 hours, every 72-96 hours, and every 7 days or after a total of approximately 12 hours of dialysis, respectively.10
t Entecavir 0.5 mg does not require dosing adjustment in patients with eCrCl ≥50 mL/min. For patients with eCrCl 30-49 mL/min, 10-29 mL/min, and ESRD on chronic hemodialysis, the dosing is 0.25 mg once daily or 0.5 mg/48 hours, 0.15 mg once daily or 0.5 mg/72 hours, and 0.05 mg once daily or 0.5 mg/7 days, respectively. Please refer to the Prescribing Information for additional dosing information for entecavir 1 mg in patients with lamivudine-refractory or decompensated liver disease.11