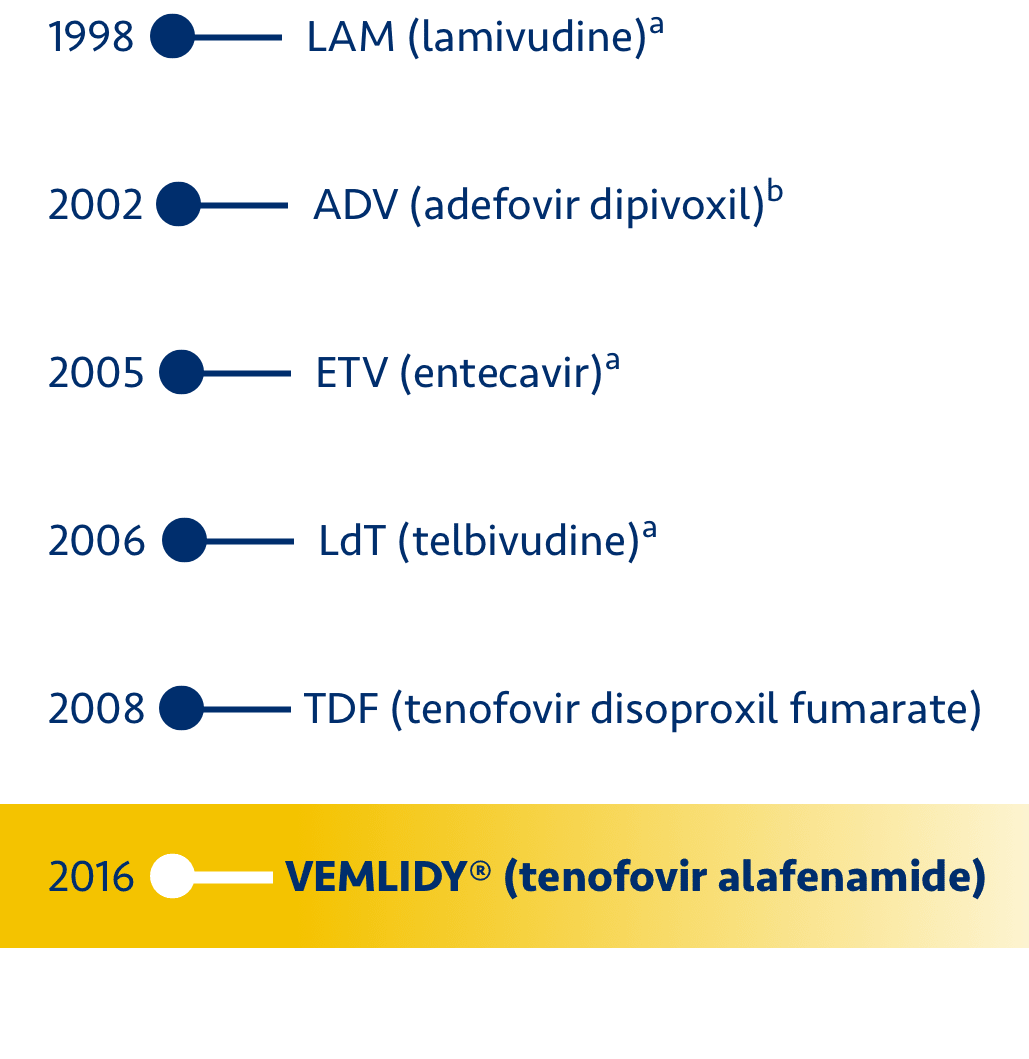
References: 1. VEMLIDY Prescribing Information. Foster City, CA: Gilead Sciences, Inc.; March 2024.
2. TDF (tenofovir disoproxil fumarate) Prescribing Information. Foster City, CA: Gilead Sciences, Inc.; April 2019.
3. BARACLUDE (entecavir) Prescribing Information. Princeton, NJ: Bristol-Myers Squibb; November 2019. 4. TYZEKA
(telbivudine) Prescribing Information. East Hanover, NJ: Novartis; January 2013. 5. EPIVIR-HBV (lamivudine)
Prescribing Information. Research Triangle Park, NC: GlaxoSmithKline; November 2018. 6. HEPSERA (adefovir dipivoxil)
Prescribing Information. Foster City, CA: Gilead Sciences, Inc.; December 2018. 7. Chan HLY, Fung S, Seto WK, et al.
Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of HBeAg-positive chronic hepatitis B
virus infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet Gastroenterol Hepatol.
2016;1:185-195. 8. Lee WA, He GX, Eisenberg E, et al. Selective intracellular activation of a novel prodrug of the
human immunodeficiency virus reverse transcriptase inhibitor tenofovir leads to preferential distribution and
accumulation in lymphatic tissue. Antimicrob Agents Chemother. 2005;49(5):1898-1906.
doi:10.1128/AAC.49.5.1898-1906.2005 9. Murakami E, Wang T, Park Y, et al. Implications of efficient hepatic delivery
by tenofovir alafenamide (GS-7340) for hepatitis B virus therapy. Antimicrob Agents Chemother. 2015;59(6):3563-3569.
doi:10.1128/AAC.00128-15 10. Agarwal K, Fung SK, Nguyen TT, et al. Twenty-eight day safety, antiviral activity, and
pharmacokinetics of tenofovir alafenamide for treatment of chronic hepatitis B infection. J Hepatol.
2015;62(3):533-540. doi:10.1016/j.jhep.2014.10.035 11. Ghany MG, Pan CQ, Lok AS, et al. AASLD IDSA Practice Guideline on treatment of chronic hepatitis B. Hepatology. 2026;83(4):974-997. doi:10.1097/HEP.0000000000001549 12. European Association for the Study of the Liver. EASL Clinical Practice
Guidelines on the management of hepatitis B virus infection. J Hepatol. 2025;83(2):502-583.
doi:10.1016/j.jhep.2025.03.018 13. Dieterich D, Graham C, Wang S, et al. It is time for a simplified approach to
hepatitis B elimination. Gastro Hep Adv. 2022;2(2):209-218. doi:10.1016/j.gastha.2022.10.004 14. You H, Wang F, Li
T, et al. Guidelines for the prevention and treatment of chronic hepatitis B (version 2022). J Clin Transl Hepatol.
2023;11(6):1425-1442. doi:10.14218/JCTH.2023.00320 15. Tong MJ, Pan CQ, Han SB, et al. An expert consensus for the
management of chronic hepatitis B in Asian Americans. Aliment Pharmacol Ther. 2018;47:1181-1200. 16. Martin P,
Nguyen MH, Dieterich DT, et al. Treatment algorithm for managing chronic hepatitis B virus infection in the United
States: 2021 update. Clin Gastroenterol Hepatol. 2021;S1542-3565(21)00818-1. doi:10.1016/j.cgh.2021.07.036