VEMLIDY—proven long-term ALT normalization in chronic hepatitis B patients with compensated liver disease1,2
Powerful efficacy maintained over the long term with 0% resistance2
ALT normalization rates at Weeks 48, 96, and 144 (2016 AASLD criteria)2,3,a,b
~75% of patients enrolled in pivotal Trials 108/110 were treatment-naïve.2
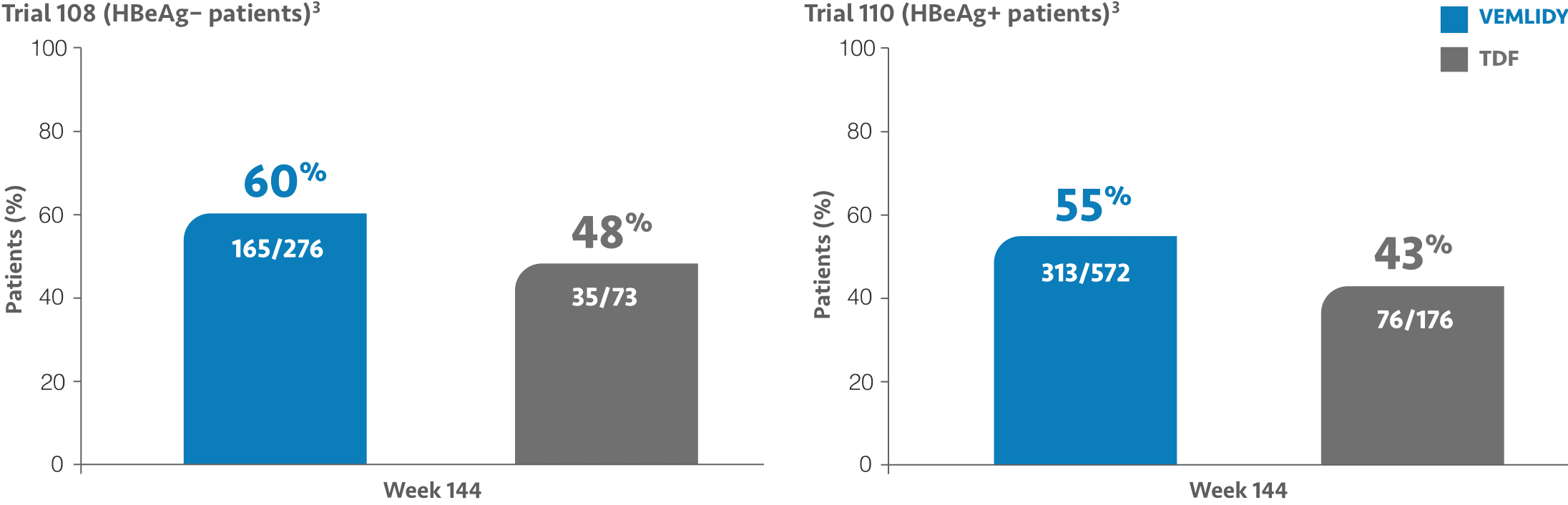

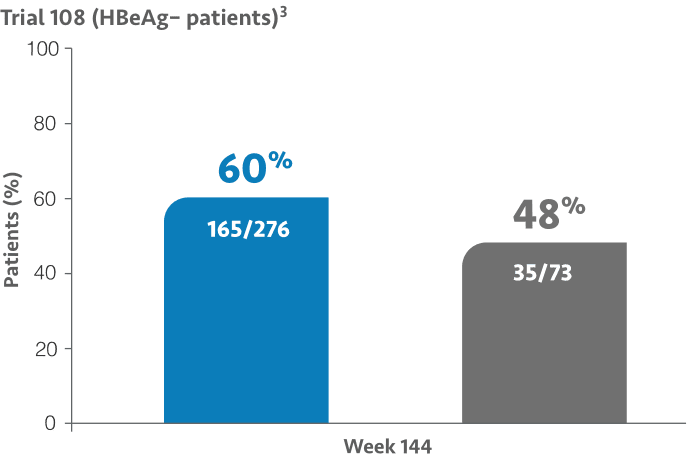
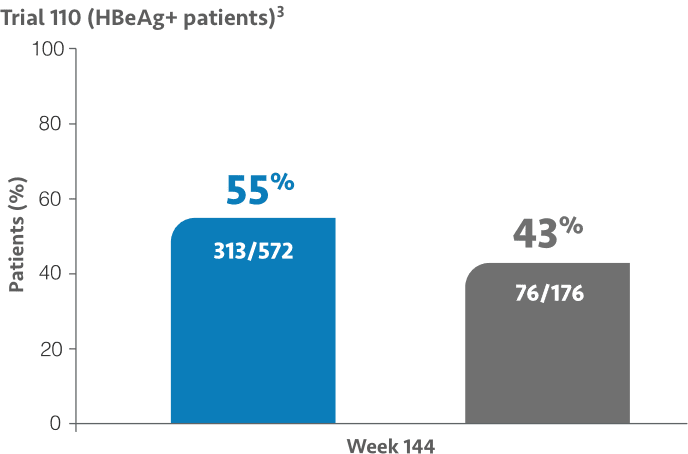
At Week 48: ALT normalization was 50% (137/276) for VEMLIDY vs 32% (44/138) for TDF in Trial 108 and 45% (257/572) for VEMLIDY vs 36% (105/290) for TDF in Trial 110.1,4-6
At Week 96: ALT normalization was 50% (139/276) for VEMLIDY vs 40% (55/138) for TDF in Trial 108 and 52% (299/572) for VEMLIDY vs 42% (121/290) for TDF in Trial 110.1,4-6
Most common adverse reactions (incidence ≥5%; all grades) in clinical studies through Week 144 were headache, upper respiratory tract infection, abdominal pain, cough, back pain, arthralgia, fatigue, nausea, diarrhea, dyspepsia, and pyrexia.1,3,4,6
AASLD=American Association for the Study of Liver Diseases; ALT=alanine aminotransferase; HBeAg=hepatitis B envelope antigen; TDF=tenofovir disoproxil fumarate; ULN=upper limit of normal.
a The population used for analysis of ALT normalization included only patients with ALT above ULN based on the AASLD 2016 criteria (>30 U/L and >19 U/L for males and females, respectively) at baseline.1
b The Week 144 analysis did not include the 66 patients from the TDF group in Trial 108 and the 114 patients from the TDF group in Trial 110 who had rolled over from double-blind TDF to open-label VEMLIDY at Week 96 prior to the study amendment.3
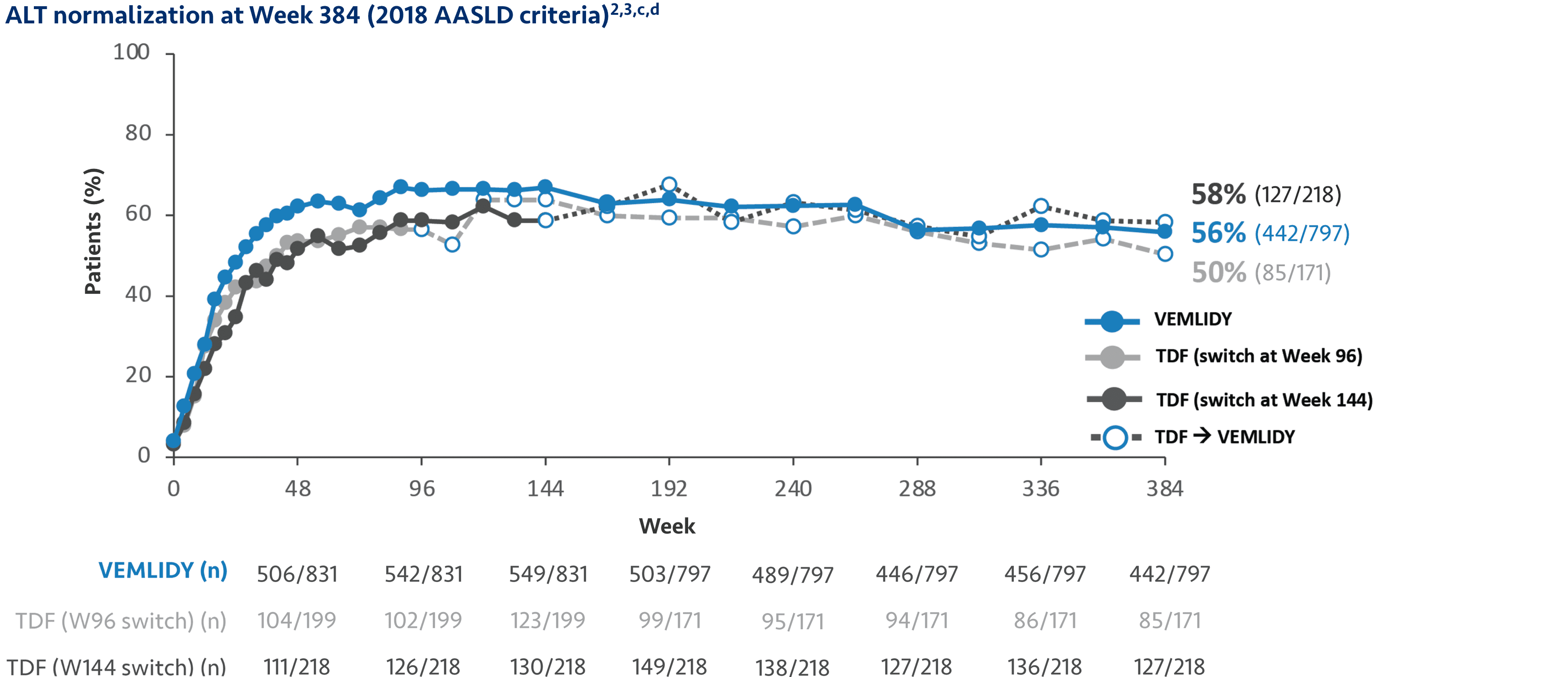
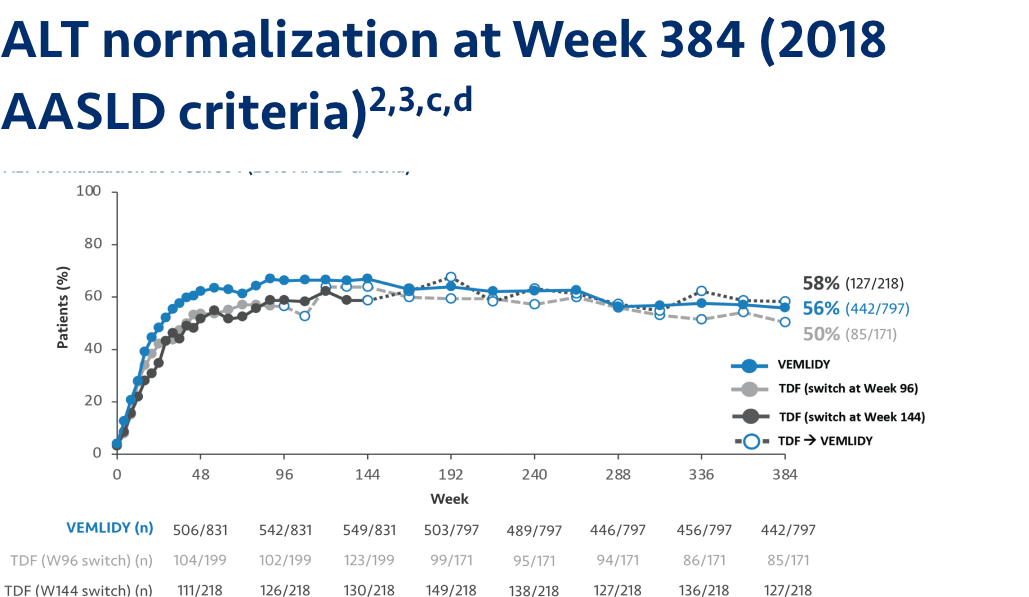
Long-term ALT normalization with VEMLIDY through 8 years
Pooled Week 384 analysis: Pooled efficacy analysis from Trials 108 and 110 was assessed at Week 384 analysis for patients in the full analysis set. This analysis includes 866 patients who continued on VEMLIDY (pooled), 207 patients who switched from TDF to VEMLIDY at Week 96, and 225 patients who switched from TDF to VEMLIDY at Week 144. Efficacy in the open-label phase was calculated using a missing=failure (M=F) patient analysis.2
Most common adverse reactions (incidence ≥5%; all grades) at Week 384 OLE were headache, upper respiratory tract infection, nasopharyngitis, arthralgia, hypertension, cough, and back pain.2
OLE=open-label extension.
c One site did not participate in protocol amendment 3, and all patients from this site (n=64) discontinued the study on or before Year 3 (Week 144); therefore, those patients who completed the planned study treatments were excluded from the M=F analysis for all visits after Year 3.2
d The population used for analysis of ALT normalization included only patients with ALT >ULN per the 2018 AASLD criteria (≤35 U/L for males and ≤25 U/L for females) at baseline.2
The 8-year analysis is not presented in the VEMLIDY full Prescribing Information.
See ALT normalization data from the switch trial.
In Trials 108 and 110, genotypic resistance analysis was performed on patients experiencing either1:
- Virologic breakthrough (2 consecutive visits with HBV DNA ≥69 IU/mL [400 copies/mL] after having been <69 IU/mL, or ≥1.0-log10 increase in HBV DNA from nadir)
- Early discontinuation at or after Week 24 with HBV DNA ≥69 IU/mL
Warnings and Precautions
- Risk of Development of HIV-1 Resistance in HBV/HIV-1 Coinfected Patients: Due to this risk, VEMLIDY alone should not be used for the treatment of HIV-1 infection. Safety and efficacy of VEMLIDY have not been established in HBV/HIV-1 coinfected patients. HIV antibody testing should be offered to all HBV-infected patients before initiating therapy with VEMLIDY, and, if positive, an appropriate antiretroviral combination regimen that is recommended for HBV/HIV-1 coinfected patients should be used.
References: 1. VEMLIDY Prescribing Information. Foster City, CA: Gilead Sciences, Inc.; March 2024. 2. Buti M, Lim YS, Chan HLY, et al. Eight-year efficacy and safety of tenofovir alafenamide for treatment of chronic hepatitis B virus infection: final results from two randomised phase 3 trials. Aliment Pharmacol Ther. 2024;60(11-12):1573-1586. doi:10.1111/apt.18278 3. Data on file. Gilead Sciences, Inc. 4. Chan HLY, Fung S, Seto WK, et al; GS-US-320-0110 investigators. Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of HBeAg-positive chronic hepatitis B virus infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet Gastroenterol Hepatol. 2016;1(3):185-195. doi:10.1016/S2468-1253(16)30024-3 5. Agarwal K, Brunetto M, Seto WK, et al; GS-US-320-0110 and GS-US-320-0108 investigators. 96 weeks treatment of tenofovir alafenamide vs. tenofovir disoproxil fumarate for hepatitis B virus infection. J Hepatol. 2018;68(4):672-681. doi:10.1016/j.jhep.2017.11.039 6. Buti M, Gane E, Seto WK, et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate for the treatment of patients with HBeAg-negative chronic hepatitis B virus infection: a randomised, double-blind, phase 3, non-inferiority trial. Lancet Gastroenterol Hepatol. 2016;1(3):196-206. doi:10.1016/S2468-1253(16)30107-8
