VEMLIDY was proven in robust, global clinical trials
The efficacy and safety profile of VEMLIDY in the treatment of adults with chronic hepatitis B infection with compensated liver disease was based on data from 3 randomized, double-blind, active-controlled trials with 1786 patients (HBeAg– and HBeAg+)1-3,a
a Study 4018 included 488 virologically suppressed patients; Study 108 included 425 HBeAg− patients; Study 110 included 873 HBeAg+ patients.
Trial designs and baseline characteristics
The efficacy and safety of switching from TDF 300 mg once daily to VEMLIDY 25 mg once daily in virologically suppressed adults with chronic hepatitis B infection were evaluated in a randomized, double-blind, active-controlled trial: Trial 4018 (N=488).1
Patients must have been taking TDF 300 mg once daily for ≥12 months, with HBV DNA less than the Lower Limit of Quantitation by local laboratory assessment ≥12 weeks prior to screening and HBV DNA <20 IU/mL at screening. Patients were randomized in a 1:1 ratio to either switch to VEMLIDY (n=243) or stay on TDF (n=245).1
At baseline, the median duration of exposure to TDF prior to the trial was similar in both treatment groups (TAF=220.0 weeks, TDF=224.3 weeks).1
At Week 48, all patients who were randomized to TDF for the controlled portion of the trial were switched to VEMLIDY for the open-label extension through Week 96.4
The primary efficacy endpoint was the proportion of patients with plasma HBV DNA ≥20 IU/mL at Week 48. Additional efficacy endpoints included the proportion of patients with HBV DNA <20 IU/mL, ALT normalization, HBsAg loss and seroconversion, and HBeAg loss and seroconversion.1
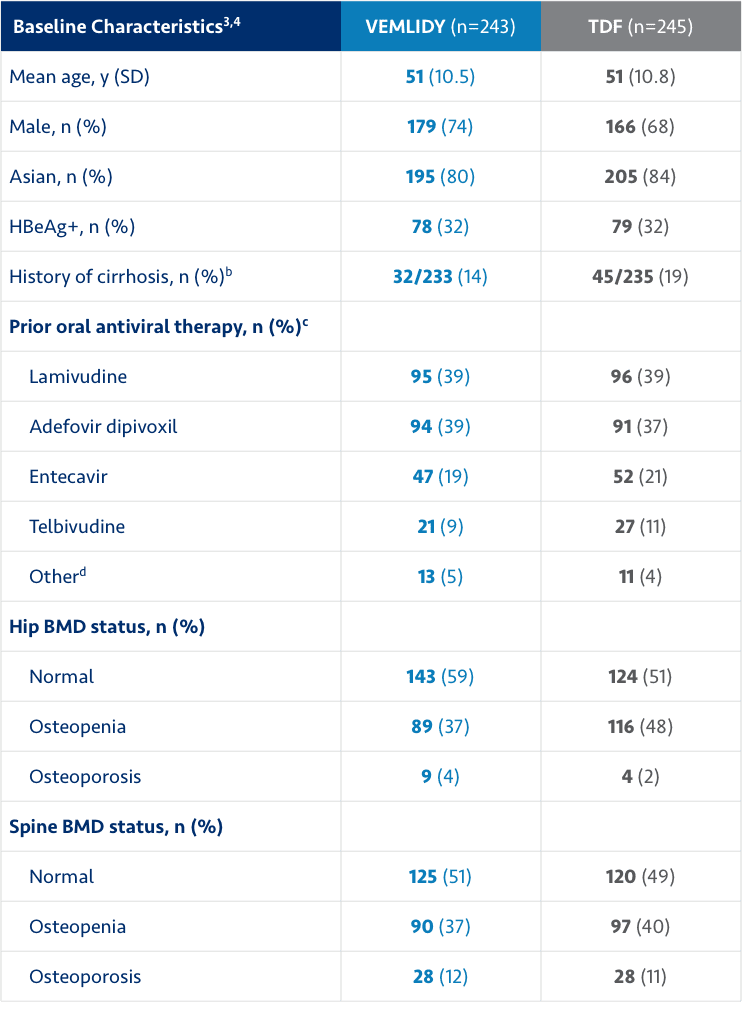
ALT=alanine aminotransferase; BMD=bone mineral density; HBeAg=hepatitis B envelope antigen; HBsAg=hepatitis B surface antigen; SD=standard deviation; TAF=tenofovir alafenamide; TDF=tenofovir disoproxil fumarate.
bExcludes patients with missing values.
cExcludes TDF.
dIncludes clevudine, emtricitabine/TDF, and TAF.3
Warnings and Precautions
- New Onset or Worsening Renal Impairment: Postmarketing cases of renal impairment, including acute renal failure, proximal renal tubulopathy (PRT), and Fanconi syndrome have been reported with TAF-containing products. Patients with impaired renal function and/or taking nephrotoxic agents (including NSAIDs) are at increased risk of renal-related adverse reactions. Discontinue VEMLIDY in patients who develop clinically significant decreases in renal function or evidence of Fanconi syndrome. Monitor renal function in all patients – See Dosage and Administration.
