VEMLIDY: A demonstrated renal safety profile through 8 years1,2
Across multiple trials, VEMLIDY demonstrated a reduced effect on renal safety parameters and improved renal function when compared to baseline1,2
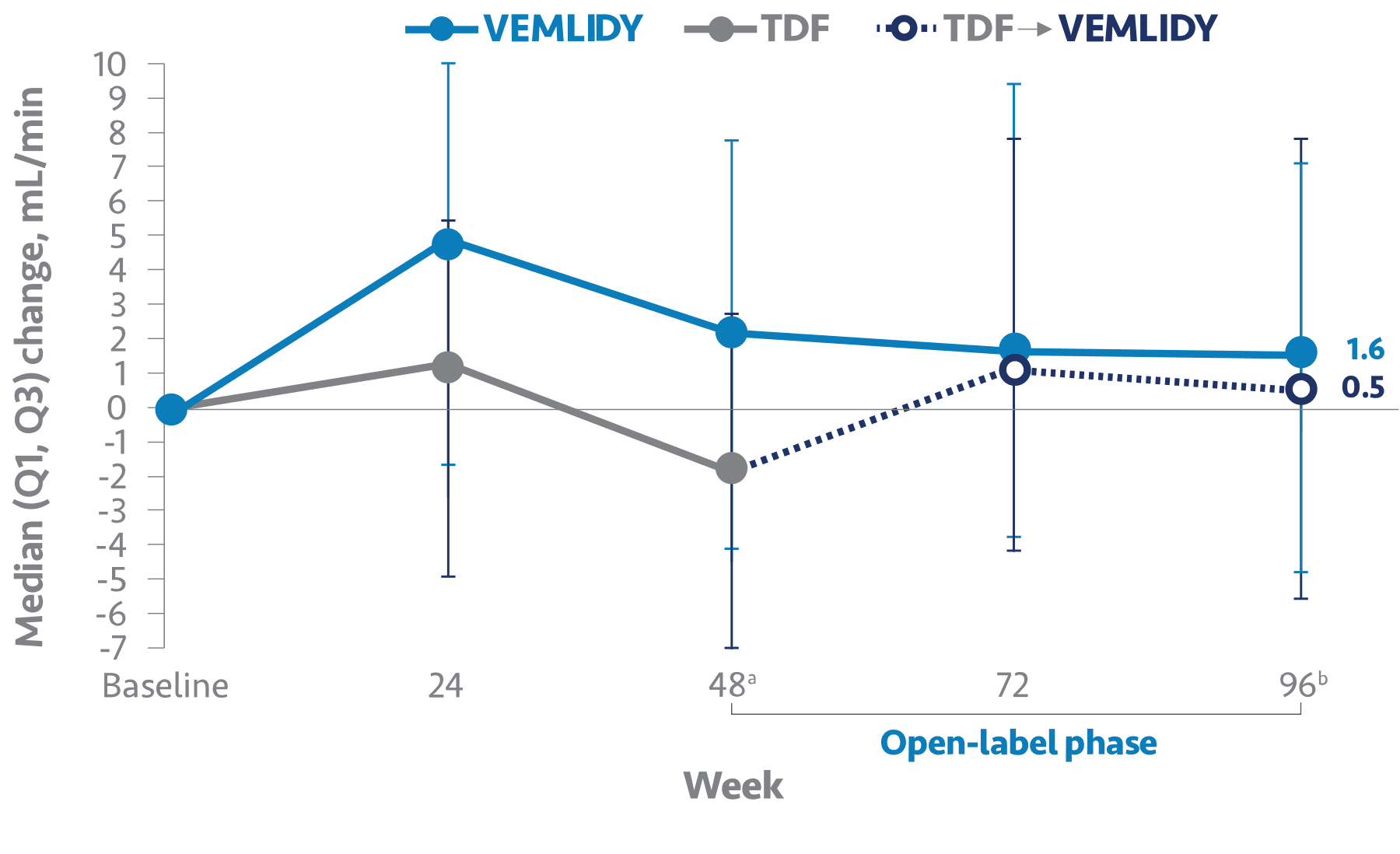
Change in eGFRCG from baseline to Week 96 (including open-label treatment after Week 48)3,4
Change in eGFRCG from baseline to Week 963
eGFRCG=estimated glomerular filtration rate by Cockcroft-Gault method, also referred to as eCrCl (estimated creatinine clearance); TDF=tenofovir disoproxil fumarate.
a P<0.0001; fallback procedure confirmed statistical significance using the adjusted alpha.3
bP=Not significant.3
- Median change from baseline to Week 48 in eGFRCG was +0.9 mL/min in the VEMLIDY group (n=243) and -2.7 mL/min in the TDF group (n=245)4
- Median baseline eGFRCG was 90.9 mL/min and 90.3 mL/min, for VEMLIDY and TDF, respectively4
The long-term clinical significance of these renal laboratory changes on adverse reaction frequencies between VEMLIDY and TDF is not known.1
Warnings and Precautions
- New Onset or Worsening Renal Impairment: Post marketing cases of renal impairment, including acute renal failure, proximal renal tubulopathy (PRT), and Fanconi syndrome have been reported with TAF-containing products. Patients with impaired renal function and/or taking nephrotoxic agents (including NSAIDs) are at increased risk of renal-related adverse reactions. Discontinue VEMLIDY in patients who develop clinically significant decreases in renal function or evidence of Fanconi syndrome. Monitor renal function in all patients – See Dosage and Administration.
